Secukinumab Treatment in Patients with Spondyloarthritis and Concomitant Demyelinating Disease: Rationale of IL-17 Inhibition and Clinical Outcome
Rossana Scrivo1, Andrea Picchianti Diamanti2*, Maria Sole Chimenti3, Emanuele Molteni1, Giorgio Sciarra1, Federico Bari2, Elisabetta Greco3, Alberto Bergamini3, Bruno Laganà2, Fabrizio Conti1
1Department of Clinical Internal, Anesthesiological and Cardiovascular Sciences, Sapienza University of Rome, Rome, Italy
2Department of Clinical and Molecular Medicine, Sapienza University of Rome, Sant'Andrea University Hospital, Rome, Italy
3Rheumatology, Allergology and Clinical Immunology, Tor Vergata University of Rome, Rome, Italy
*Corresponding Author: Andrea Picchianti Diamanti, Department of Clinical and Molecular Medicine, Sapienza University of Rome, Sant'Andrea University Hospital, Rome, Italy.
Received: 03 August 2022; Accepted: 23 August 2022; Published: 05 October 2022
Article Information
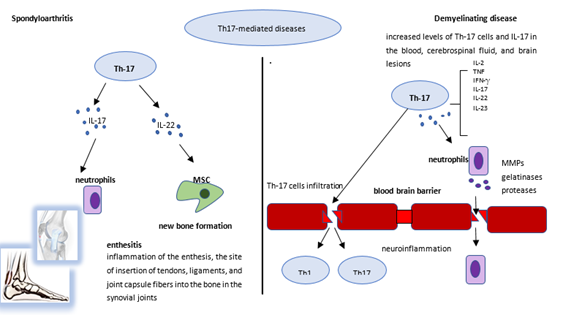
Citation: For patients with spondyloarthritis (SpA), the coexistence of demyelinating diseases complicates the choice of the therapeutic agent, as demyelinating diseases should not be targeted with tumor necrosis factor inhibitors. However, targeting interleukin (IL)-17 may have a beneficial effect on both diseases. Secukinumab is a monoclonal antibody that selectively binds to and inhibits IL-17A. To date, there have been only a handful of case studies reporting the use of secukinumab in patients with concomitant SpA and multiple sclerosis (MS), showing good efficacy and safety profiles. Here, we focus on the rationale of inhibiting IL-17 with secukinumab in the setting of concomitant SpA and demyelinating diseases, also discussing the clinical outcome experienced in patients. Currently, secukinumab seems the only safe treatment for patients with concomitant SpA and MS. <p>Biological Disease-Modifying Antirheumatic Drugs; Demyelinating Diseases; Multiple Sclerosis; Secukinumab; Spondyloarthritis</p> Spondyloarthritis (SpA) is an umbrella term that covers a group of inflammatory diseases sharing pathogenetic and clinical features. Recently, SpA has been classified into two main groups: axial SpA, including ankylosing spondylitis (AS) and non-radiographic axial SpA, which predominantly affects the sacroiliac joints and spine, and peripheral SpA, including psoriatic arthritis (PsA), reactive arthritis, arthritis related to inflammatory bowel disease, and undifferentiated SpA [1-3]. Among rheumatologic diseases, SpA is quite common, with an incidence ranging from 0.48 to 63/100.000 persons/year and an East-West gradient [4]. Prevalence rates vary between 0.2% in Europe and North America and 0.02% in the Sub-Saharan population [5]. Axial SpA recognizes a multifactorial pathogenesis, where the genetic predisposition can favor an altered inflammatory response to external triggers such as gut dysbiosis and biomechanical stress [6, 7]. As a result, lymphocytes produce dysregulated levels of pro-inflammatory cytokines, mainly tumor necrosis factor (TNF) and interleukin (IL)-2 and 6, but the crucial role is played by the IL-12/23 and IL-17 axis [3, 8]. These molecules drive inflammatory damage involving the sacroiliac joints and the spine with the production of syndesmophytes and the calcification of the enthesis [9]. Patients with SpA suffer from inflammatory back pain, peripheral arthritis, and enthesitis, and may present extra-articular manifestations such as anterior uveitis and inflammatory bowel disease [10]. According to the recommendations by the European Alliance of Associations for Rheumatology (EULAR) [11], the first therapeutic line for axial SpA is a combination of physical exercise and non-steroidal anti-inflammatory drugs (NSAIDs) at full dose for at least four weeks. The second-line drugs are the biotechnological drugs or biological disease-modifying anti-rheumatic drugs (bDMARDs), such as TNF inhibitors (TNFi). Recently, the possibility of targeting IL-17 has widened the therapeutic options in SpA [1-4]. Indeed, the availability of bDMARDs targeting different molecules allows the therapeutic strategy to be directed in a more personalized manner, based on the presence of extra-articular manifestations as well as comorbidities. Furthermore, the availability of these drugs has led different international working groups to develop and validate standardized tools for the assessment of their efficacy. In patients with axial SpA, the disease activity can be monitored over time by using the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) or the Ankylosing Spondylitis Disease Activity Score (ASDAS). BASDAI was developed in the British city of Bath in 1994 as a self-reported questionnaire encompassing 6 items that explore aspects of fatigue, pain, discomfort, and morning stiffness on a 0-10 visual analog scale. A global score of 4 or more defines an active disease [12]. On the other hand, ASDAS, developed in 2009 by the Assessment in Spondyloarthritis International Society (ASAS) working group, is a composite score, based on patient’s reported assessments of back pain, peripheral joint pain/swelling, duration of morning stiffness together with general well-being and markers of inflammation (C-reactive protein: CRP or erythrocyte sedimentation rate: ESR). Four disease activity states were identified: “inactive disease”, “low disease activity”, “high disease activity” and “very high disease activity”, separated by 3 cut-off values (<1.3, <2.1, and >3.5) [13, 14]. Among the group of peripheral SpA, PsA is the most common [3]. Again, PsA is a multifactorial disease, in which the genetic background triggers the main risk factor for PsA onset, represented by a personal history of psoriasis (PsO) or a first-degree familiar affected [15]. Furthermore, biomechanical stress and infections may be involved, all stimulating dendritic cells and polymorphonuclear cells to release self-peptides such as LL-37, human defensins, and lysozyme [16]. On the other hand, polymorphonucleocytes release TNF and neutrophil extracellular traps (NETs), which are a web of DNA, RNA, and proteins aimed at protecting against infections, but also implicated in the pathogenesis of a growing number of immune-mediated conditions [16]. In patients with PsA, these mediators promote the activation of the IL-12/23 axis and the differentiation of the T helper lymphocytes into Th-17 cells and gd lymphocytes producing great amounts of IL-17 [16]. This inflammatory response has its focus in the enthesis, the site of insertion of tendons, ligaments, and joint capsule fibers into the bone in the synovial joints [17]. Apart from enthesitis, other clinical manifestations in PsA include peripheral arthritis (which is usually oligoarticular and asymmetric but can also be polyarticular and symmetric mimicking rheumatoid arthritis), spondylitis, and dactylitis [18]. The clinical presentation differs between females and males, because women usually show a polyarticular pattern with higher rates of fatigue, pain, and physical and work disability [19]. The therapeutic approach for PsA should be driven by the main site involved, as suggested by the EULAR and the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) recommendations [20, 21]. Usually, the first line is represented by conventional synthetic DMARDs (csDMARDs) including methotrexate, sulfasalazine, cyclosporine, and leflunomide. From the new millennium, other agents have enriched the therapeutic armamentarium, including bDMARDs targeting TNF, IL-12/IL-23, IL-17, and small synthetic molecules inhibiting the Janus kinase pathway or the phosphodiesterase-4 activity [22, 23]. To monitor disease activity and quantify therapeutic efficacy in PsA, a great variety of scores have been published [24]. The Disease Activity in PSoriatic Arthritis (DAPSA) is the only unidimensional score, meaning that merely the musculoskeletal involvement is considered. DAPSA derives from the arithmetic sum of patient-reported pain and global assessment on a 0–10 scale, joint involvement signified by 66 swollen joint counts and 68 tender joint counts, and acute phase response, represented by CRP (mg/dl). The final score identifies different activity states: remission (0-4), low disease activity (5-14), moderate disease activity (15-28), and high disease activity (>28) [25]. As said above, SpA is often accompanied by several comorbidities, such as osteoporosis, cardiovascular diseases, anxiety and depression, and fibromyalgia [26]. Although rare, one of the most challenging comorbidities in patients with SpA is represented by demyelinating diseases, which should not be targeted with TNF inhibitors [27], whereas IL-17 neutralization could have a beneficial effect [28]. The most common demyelinating disease is multiple sclerosis (MS), an inflammatory and neurodegenerative chronic condition of the central nervous system (CNS) characterized by focal areas of demyelination, inflammation, and glial reaction easily detectable by magnetic resonance imaging (MRI) [29, 30]. The incidence of MS varies among continents and seems to have arisen in the last decades [31]. In Europe, the incidence varies from 7.2 to 12.2/100,000 persons/year with the highest rates in Scotland, Northern Ireland, and Sardinia. In the Americas, the highest rates are in Canada (23.9/100,000 persons/year, Alberta) and the lowest in South America (Argentina 1.4/100,000 persons/year) [32]. Traditionally, the histopathologic hallmark of brain inflammation was the CD4+ T-lymphocyte infiltrate, but in recent years the role of CD8+ T-cell and CD17+ lymphocytes has emerged. Cells expressing the CD17+ phenotype have been identified in active MS lesions and their suppression has been demonstrated to reduce the number of active lesions and send the disease to remission [28, 33]. Furthermore, increased levels of Th-17 cells and IL-17 have been observed in the blood, cerebrospinal fluid (CSF), and brain lesions which correlate with disease activity and severity [34]. Even elevated TNF levels have been found in the serum, CSF, and brain plaques in patients with MS, where the TNFR1 signaling mediates blood-brain barrier (BBB) disruption, demyelination, and glutamate-excitotoxicity, while TNFR2 signaling mediates immune modulation via regulatory T cells (a particular subgroup of lymphocytes) activation-expansion, tissue regeneration, and neuroprotection [35]. The known implication of TNF and IL-17 in SpA and MS is summarized in Table 1, while Figure 1 illustrates the role of Th-17 cells in the pathogenesis of both diseases. TNF TNF inhibitors IL-17 IL-17 inhibitors Spondyloarthritis Elevated levels in the serum and joints TNFR1 signaling= Main mediator of early inflammation and tissue catabolism TNFR2 signaling= Anti-inflammatory action, could mediate tissue anabolism (new bone formation in transgenic mice) Demonstrated safety and effectiveness in several clinical trials and real-life studies and are approved for the treatment of both AS and PsA Increased levels of IL-17A and Th-17 cells in peripheral blood, joints, and psoriatic lesions Polymorphisms in genes affecting the generation of Th-17 cells altered susceptibility to the disease Demonstrated safety and effectiveness in several clinical trials and real-life studies and are approved for the treatment of both AS and PsA Multiple sclerosis Elevated levels in the serum, CSF, and brain plaques TNFR1 signaling= Mediates BBB disruption, demyelination and glutamate-excitotoxicity TNFR2 signaling = Mediates immune modulation via Treg activation-expansion, tissue regeneration, and neuroprotection Not recommended since reactivation or new onset of MS have been described. Block of TNFR2 signaling can induce CNS inflammation and demyelination. Upregulation of TNF expression in the CNS (anti-TNF cannot penetrate BBB) Triggering of latent infection Increased levels of Th-17 cells and IL-17 in the blood, CSF, and brain lesions Correlation with disease activity and severity By impairing tight junctions in the BBB, IL-17 infiltrates CNS and induces neuroinflammation and damage Secukinumab was safe and effective in treating RRMS in a proof-of-concept clinical trial It also showed no MS reactivation in patients with concomitant SpA/MS Table 1: TNF and IL-17 in spondyloarthritis and multiple sclerosis. Abbreviations: AS- Ankylosing Spondylitis; BBB- Blood-Brain Barrier; CNS- Central Nervous System; CSF- Cerebrospinal Fluid; IL- Interleukin; MS- Multiple Sclerosis; PsA- Psoriatic Arthritis; RRMS- Relapsing-Remitting Multiple Sclerosis; SpA- Spondyloarthritis; Th-17- T Helper 17 Cells; TNF- Tumor Necrosis Factor; TNFR1- Tumor Necrosis Factor Receptor 1; TNFR2- Tumor Necrosis Factor Receptor 2; Treg- Regulatory T Cell. Figure 1: The role of Th-17 cells in the pathogenesis of spondyloarthritis and demyelinating disease. In patients with spondyloarthritis (left), the enthesis is the focus of the inflammatory response. Th-17 cells produce a great amount of IL-17 and other cytokines that induce the production of a variety of other soluble mediators by resident mesenchymal cells, which can trigger neutrophil migration and activation, thus amplifying the inflammatory response in the enthesis. Furthermore, IL-17, together with other inflammatory molecules such as IL-22, stimulates the activation and proliferation of resident mesenchymal stem cells (MSCs) from the perientheseal periosteum which ultimately leads to new bone apposition. In patients with demyelinating disease (right), neutrophils activated by IL-17 and other soluble mediators released by Th-17 cells produce several enzymes such as matrix metalloproteinases (MMPs), gelatinases, and proteases. In turn, these may efficiently disrupt the blood-brain barrier leading to neuroinflammation and damage through several mechanisms. Clinically, MS is characterized by variable phenotypes: relapsing-remitting MS (RRMS), primary progressive MS (PPMS), and secondary progressive MS (SPMS) [36]. These different phenotypes underlie different pathogenetic pathways and, consequently, need different disease-modifying therapies (DMTs). The therapeutic armamentarium is composed of five injectable agents (glatiramer acetate, interferon-β-1a, and interferon-β-1b) and two oral agents (teriflunomide and dimethyl fumarate). In addition, for the remitting forms, two monoclonal antibodies have been approved: ocrelizumab (a humanized monoclonal antibody against the CD20 molecule) and natalizumab (an inhibitor of α4β1 integrin) [37-39]. To date, 12 patients with concomitant SpA and MS treated with secukinumab, which selectively binds to and inhibits IL-17A, have been published, all showing good efficacy and safety profiles [40-45]. Among these, 3 cases have been published by our groups, two in 2019 and one in 2021 [41, 42]. Briefly, our cases comprise a female patient [41] affected by RRMS and AS since 2016 and successfully treated with secukinumab 150 mg from October 2017 until 2019, when the case was published. However, in 2020 a neurological relapse led to stopping the treatment with secukinumab and starting cladribine. On this new treatment regimen, MS symptoms and RM signs improved, but the patient experienced a worsening in AS activity, and in February 2021 secukinumab was reintroduced, leading to a state of persistent remission. The second patient [41] was a female affected by AS since 2012 and treated with adalimumab (a TNF-inhibitor) till 2017 when she received a diagnosis of MS-like syndrome. Adalimumab was immediately stopped, and the patient was treated with glucocorticoids and then with dimethyl fumarate. Because of poor response, secukinumab was added, and in January 2019, after 18 months, there was neither evidence of MS-like activity nor of AS activity. Unfortunately, the patient was lost to follow-up. The last patient [42] was a male with AS who developed MS while treated with adalimumab after failing etanercept (another TNF-inhibitor). Adalimumab was stopped and secukinumab 150 mg was started in 2018. After 9 months, following an exacerbation of AS activity, the dosage of secukinumab was increased to 300 mg with a good clinical response in both diseases. Here we report 5 new patients with coexistent SpA and demyelinating disease, all longitudinally observed after starting treatment with secukinumab, with outcomes assessed in terms of effectiveness and safety for both diseases. Informed consent was obtained from all subjects involved in the study. In addition, the relevant literature is discussed. For the literature review, a search of the PubMed database and ClinicalTrials.gov was conducted using several interrelated queries (e.g., “psoriatic arthritis” OR “Spondyloarthritis” OR “ankylosing spondylitis” OR “psoriasis”; “secukinumab” AND “multiple sclerosis” OR “demyelinating disorders”). Only English-language publications were selected. The search results were then supplemented by browsing the reference lists of identified articles, and by including other documents (abstracts/oral presentations) suggested by the authors’ experience. The search was updated on May 3, 2022. 3.1 Patient 1 The first patient is a 66-year-old female with a personal history of breast cancer and depression. In 1971, at the age of 16, she had suffered from retrobulbar optic neuritis and in 1996 received a diagnosis of MS after a new episode of optic neuritis. She successfully started therapy with IFN-β (Betaferon), however, this was suspended in 2004 due to low compliance. In May 2005, an MRI in T2- and FLAIR-weighted images showed hyperintense lesions in the corona radiata, centrum semiovale, and periventricular white matter. The spinal cord was unharmed. In 2006, IFN-β 1A (Avonex) was started, which achieved good symptom control and no progression of cerebral lesions until October 2014, when an MRI demonstrated a new T2-weighted hyperintense lesion in the cervical tract of the spinal cord. Nonetheless, IFN-β 1A was not stopped until June 2016, when the patient developed leukopenia, dullness, and asthenia. Concomitantly, the patient experienced migratory arthralgias in the hands, which were treated with nimesulide. In December 2017, a new lesion in the frontal right subcortical white matter was detected on brain MRI, and IFN-β 1A was reintroduced. In October 2018, because of arthritis in the small joints of the hands, a positive familial history of PsO, multiple tenosynovitides detected at ultrasound imaging of the joints, and no autoantibody found in laboratory tests, the patient received a diagnosis of PsA (DAPSA 28.9). Apremilast was started in October 2018 (DAPSA after one month 4.72, after 3 months 1.24). In October 2019, because of PsA relapse (DAPSA 20), the rheumatologist switched treatment to secukinumab 150 mg/month, and remission was achieved (DAPSA 0.4) in July 2020. In 2021, the patient complained of paresthesia in the arms and an MRI showed a new area of hyperintensity in T2-weighted images without contrast enhancement in the spinal cord at the C2-C3 level. As the patient’s PsA was still in remission (DAPSA 2.1) in October 2021, the clinicians decided to continue secukinumab, with ongoing monitoring of both laboratory assessments and neurological imaging. 3.2 Patient 2 The second patient is a male aged 61 years. In 2005, at the age of 45, he developed low back pain, sciatica, and alternating buttock pain. A rheumatologist diagnosed AS (ASDAS 7.3) based on these clinical manifestations and pelvic MRI findings (T2-hyperintensity of the right sacral ala and erosions in the left sacral ala). Treatment with methotrexate was started and, after 6 months, sulfasalazine was added for the persistence of LBP and the elevation of inflammatory indexes (ASDAS-ESR 3.14). Because no amelioration was achieved, etanercept was started in June 2009 (ASDAS-ESR after one month 1.39). In March 2018, the patient experienced paresthesia in both feet and hands. An MRI revealed multiple and defined areas of alteration of the signal, as for inflammatory lesions, in T2-weighted images in both cervical and dorsal spinal cord, and multiple areas of hyper-intensity in the periventricular white matter, the corona radiata, and the centrum semiovale in STIR and T2 sequences in the brain. A neurologist diagnosed MS, and, in October 2018, the patient started secukinumab 150 mg/month. In July 2021, AS was in remission (ASDAS-ESR 1.2), and the neurologic lesions were unchanged on MRI. 3.3 Patient 3 The third patient is a 31-year-old man who has been affected by PsO since 2003 and PsA since 2009 with oligoarticular involvement. He has been treated over the years with NSAIDs, glucocorticoids, cyclosporine, and methotrexate, with clinical efficacy only on cutaneous manifestations. To obtain control over arthritis, the rheumatologist implemented methotrexate therapy by adding etanercept, which was suspended after 6 months due to a diagnosis of MS. Dimethyl fumarate was started with a good clinical response. In January 2020, due to the presence of PsO and because of PsA activity (DAPSA 19), treatment with secukinumab 300 mg/month was started with a good clinical response on both skin and joint manifestations, achieving a low disease activity (DAPSA 5) associated with MS stability in July 2020. 3.4 Patient 4 The fourth patient is a 53-year-old female, who was diagnosed with RRMS in 2001 and sequentially treated with azathioprine, mycophenolate, glatiramer acetate, and IFN-β 1A. In 2008, the patient was diagnosed with axial PsA and treated, over the following 10 years, with sulfasalazine, NSAIDs, and low-dose glucocorticoids with good clinical control. In February 2019, a brain MRI showed small high-signal areas in the white matter without post-contrast enhancement. One month later, due to a progressive worsening of disease activity (BASDAI 8.1), the rheumatologist decided to start secukinumab at 150 mg/month. However, secukinumab was interrupted after 6 months due to primary failure (BASDAI 7.1). Neurological relapse was not reported during the follow-up period. 3.5 Patient 5 The last patient is a 59-year-old female, who suffered from RRMS from the age of 22 and was treated with IFN-β 1A for several years. In November 2008, she received a diagnosis of PsA with peripheral involvement, initially treated with methotrexate (stopped after 5 years for gastrointestinal intolerance), then cyclosporin (suspended after 6 months for hypertension), and low-dose glucocorticoids. In September 2019, a brain MRI highlighted multiple high signal areas in the white matter and C2 (without post-contrast enhancement). One month later, the rheumatologist started secukinumab at 150 mg/month plus low-dose prednisone. At that time, the patient’s DAPSA score was 27.4, but it gradually lowered to 13 after 12 months, and to 8.5 after 18 months. RRMS appeared to be under control and in March 2021, another brain MRI showed no further inflammatory lesions. The demographic and clinical characteristics of the 5 patients described are summarized in Table 2. Table 2: Demographic and clinical characteristics of the patients treated with secukinumab for concomitant SpA and demyelinating disease. Abbreviations: ADA- Adalimumab; AS- Ankylosing Spondylitis; ASDAS- Ankylosing Spondylitis Disease Activity Index; BASDAI- Bath Ankylosing Spondylitis Disease Activity Index; DAPSA- Disease Activity In Psoriatic Arthritis; DD- Demyelinating Disease; ETN- Etanercept; F- Female; FU- Follow-Up; GOL- Golimumab; M- Male; MS- Multiple Sclerosis; PsA- Psoriatic Arthritis; Rh- Rheumatological; RRMS- Relapsing-Remitting Multiple Sclerosis; TNF- Tumor Necrosis Factor; yrs- Years. * BASDAI/ASDAS for patients with AS. ** DAPSA for patients with PsA. † Before the onset of secukinumab. ‡ Last observation during treatment with secukinumab. δ For case no. 2 only BASDAI is available. Herein we provide the rationale for the use of IL-17i in patients with concomitant SpA and demyelinating disease, also describing the clinical outcome. Growing evidence suggests that SpA and MS share some immunopathogenic pathways, including dysregulation in the Th-17 cell axis. Th-17 cells are a small proportion of functionally heterogeneous CD4+ cells (1%–3%), comprising subpopulations that secrete IL-17 [46]. Th-17 and cytotoxic T cells are the primary sources of IL-17A, which may also be locally released by cells of the innate immunity [47]. IL-17A, via the activation of NF-κB and stabilization of the mRNA of inflammatory mediators, can induce the production of pro-inflammatory cytokines, promote neutrophil adhesion, and activate microglial cells to secrete other inflammatory molecules such as IL-1β, TNF, and IL-6. In addition, Th-17 cells can induce apoptosis in neurons by Fas/FasL interaction [48]. It is well known that Th-17 cells play a pivotal role in SpA onset and maintenance. Indeed, increased levels of IL-17A and Th-17 cells were shown in peripheral blood of SpA patients, as well as in peripheral joints, axial skeleton, and psoriatic lesions [49-53]. Also, solid evidence for the role of Th-17 cells in MS is sustained by the capability of these cells to infiltrate the CNS by impairing tight junctions in the BBB and induce neuroinflammation and damage through several mechanisms [48]. Increased levels of mononuclear cells expressing IL-17A and proinflammatory cytokines, such as IL-2, TNF, IFN-γ, IL-17, IL-22, and IL-23, deriving from Th-1 and Th-17 cells have been found in the blood and CNS of progressive MS patients [54-56]. In addition, IL-17-producing T cells seem to correlate with plaque activity, with over 70% observed in active MS lesions and only 17% in inactive plaques [33]. Secukinumab is a human IgG1κ monoclonal antibody that inhibits IL-17A [47] and is approved to treat PsO, PsA, and axial SpA. Importantly, secukinumab is the only bDMARD licensed for the treatment of AS, other than TNF inhibitors. For patients with PsA, in addition to secukinumab and anti-TNF agents, the therapeutic strategy includes ixekizumab, another anti-IL-17 agent, and ustekinumab, an IL12/23 monoclonal antibody. The choice among these bDMARDs is substantially empirical. Thus, to tailor the therapeutic agent and optimize the safety and effectiveness in patients, the presence of comorbidities must be considered. When SpA and demyelinating disease are simultaneously present, treatment with TNF inhibitors is not recommended [57] since demyelination of the central and peripheral nervous system has been associated with the use of these agents [27, 58-60]. To explain this association, several hypotheses have been proposed [61]. The first is the so-called lack of entry hypothesis. In this model, the focus is on the BBB, which allows chemokines but not immunoglobulins and, therefore, bDMARDs, to pass. Hence, TNF locally produced by the autoreactive T cells can damage the brain, since TNF inhibitors cannot hamper it. The second hypothesis is focused on the autoreactivity of T cells because the lack of inhibition of local TNF does not explain the worsening of MS under anti-TNF treatment. It has been hypothesized that anti-TNF agents can implement the number of autoreactive T cells, as seen in animal models. Due to the inflammatory status in the brain vessels caused by active MS, these lymphocytes can more easily pass the BBB, mediating and enhancing the local immune response and therefore worsening the MS status. The other two theories consider an altered balance in cytokine production, with a reduction of IL-10 and an increase of IL-12 and IFN-γ, and the so-called “sponge effect” of TNF inhibitors, capable of driving TNF out of joints and bowel, but not from the brain due to the BBB. Finally, the last theory states that TNF inhibition could precipitate a latent infection, critical for the development of MS [61]. On the other hand, secukinumab was shown to be effective in treating RRMS in a proof-of-concept clinical trial of 73 patients randomized to receive intravenous secukinumab 10 mg/kg or placebo at weeks 0, 2, 4, 8, 12, 16, and 20 [28]. Secukinumab not only reduced (albeit without reaching statistical significance) the number of combined unique active lesions by 49% but significantly lowered the mean cumulative number of new gadolinium-enhancing T1 lesions by 67% (p=0.003) at week 24 [28]. These encouraging results associated with the above reported immunopathogenic rationale in demyelinating diseases, together with the known effectiveness of secukinumab in SpA patients, provide the basis for its adoption in patients with concomitant SpA and MS. In our cohort, the 2 patients who firstly received a diagnosis of SpA (one with AS and the other with PsA) were taking a TNF inhibitor (etanercept in both cases) while developing MS. Overall, treatment with secukinumab led to remission of both SpA and MS. Secondary inefficacy was observed in patient 1, and a lack of response was reported in patient 4. Therefore, secukinumab was effective against SpA but did not allow complete control of MS, since 20% of our patients needed other therapies to achieve neurological remission. In a recent literature search, 8 cases of concomitant SpA and MS (3 of which have previously been described by us and reported above with a longer follow-up for 2 of them) plus 4 additional patients were described [62]. Thus, including our new cases, a total of 17 patients with concomitant SpA and MS treated with secukinumab have currently been reported. After secukinumab therapy, all but one patient presented a significant improvement in AS and PsA involvement, generally evaluated through DAPSA, BASDAI, and ASDAS, with clinical remission reached in most of them. Regarding neurologic involvement, one patient had a progression of MS symptoms and brain lesions, 13/17 (76%) patients remained in stable conditions, whereas 3/17 (18%) patients showed improvement in MRI lesions. Ten patients were affected by AS and 7 by PsA. One AS patient needed an off-label secukinumab dosage of 300 mg every 4 weeks due to secondary failure of the standard 150 mg dose [62]. The follow-up period ranged from 6 to 36 months. Eight out of seventeen (47%) patients received anti-TNF therapy before MS onset. Secukinumab was used as a first-line biologic in 9/17 (53%) patients. Most patients underwent glucocorticoids and IFN-β as MS therapy before starting secukinumab. In patients with concomitant SpA and demyelinating disease, the pathogenetic pathways seem to converge on IL-17, thus its inhibition could offer an important treatment alternative to TNF blockers that are contraindicated. Here we provided the rationale for the use of IL-17 inhibitors in these patients and reported 5 new cases longitudinally observed after starting treatment with secukinumab, together with a discussion of the relevant literature. Overall, this therapeutic agent showed strong effectiveness against SpA joined to a good safety profile on MS. Currently, secukinumab seems the only treatment which may be safely administered in patients with concomitant SpA and MS. Editorial services were provided by Melanie Gatt (Ph.D.), an independent medical writer, on behalf of Health Publishing & Services Srl. This unconditional support was funded by Novartis Farma. This research received no external funding. The authors declare no conflict of interest. Conceptualization, R.S.; data curation, F.B., E.G. and A.B.; writing—original draft preparation, R.S., A.P.D., M.S.C., E.M., and G.S.; writing—review and editing, B.L. and F.C.; supervision, F.C. All authors have read and agreed to the published version of the manuscript. Informed consent was obtained from all subjects involved in the study.Abstract
Keywords
Article Details
Introduction
2. Patients and Methods
3. Results
![]()
4. Discussion
5. Conclusions
Acknowledgments
Funding
Conflicts of Interest
Author Contributions
Informed Consent Statement
References


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
