A Case of Long-Term Survival with Own Liver in the Patient of Biliary Atresia Who Developed Hepatocellular Carcinoma
Yasunari Tanaka1, Kouji Masumoto1, Kazunori Ishige2, Kuniaki Fukuda2, Toko Shinkai1, Hajime Takayasu1*
1Department of Pediatric Surgery, Faculty of Medicine, University of Tsukuba, Ibaraki, Japan
2Department of Gastroenterology, Faculty of Medicine, University of Tsukuba, Ibaraki, Japan
*Corresponding Author: Dr. Hajime Takayasu, Department of Pediatric Surgery, Faculty of Medicine, University of Tsukuba, Ibaraki, Japan
Received: 16 December 2019; Accepted: 26 December 2019; Published: 30 January 2020
Article Information
Citation: Yasunari Tanaka, Kouji Masumoto, Kazunori Ishige, Kuniaki Fukuda, Toko Shinkai, Hajime Takayasu. A Case of Long-Term Survival with Own Liver in the Patient of Biliary Atresia Who Developed Hepatocellular Carcinoma. Archives of Clinical and Medical Case Reports 4 (2020): 110-118.
View / Download Pdf Share at FacebookAbstract
Here we report a case of long-term survival with own liver in the patient of biliary atresia (BA) who developed hepatocellular carcinoma (HCC). The patient was a 34-year-old man. He underwent a portal jejunostomy with a diagnosis of BA (IIIb1ν) on the 94th day after birth, with a favorable post-operative course. Contrast-enhanced CT upon admission for cholangitis at the age of 34 suspected HCC, with an AFP level of 1.3 ng/ml and increased PIVKAII to 1,199 mAU/ml. He was diagnosed with HCC on MRI, and the tumor embolus had spread to the left branch of the portal vein; thus, resection and transplantation were considered contraindication. Proton therapy was performed, but he died 11 months after the detection of HCC. To date, 26 cases of HCC including this one have been reported, of which 20 were those in minors. Many of the patients with early onset have poor prognosis such as poor reduction of jaundice, while many of those with adult onset have relatively favorable prognosis as in this case; therefore, it was suggested that the progression of cholestatic cirrhosis is associated with carcinogenesis.
Keywords
<p>Biliary atresia; Hepatocellular carcinoma; Long-term survivors</p>
Article Details
Abbreviations:
BA - Biliary Atresia; HCC - Hepatocellular Carcinoma; CT - Computed Tomography; MRI - Magnetic Resonance Imaging; PBC - Primary Biliary Cirrhosis; LT – Liver Transplantation
1. Introduction
Biliary atresia (BA) is rarely considered to be associated with hepatocellular carcinoma (HCC) [1]. However, it is imaginable that cholestatic cirrhosis progresses slowly, which increases the risk of carcinogenesis. Therefore, it is expected that the patients who develop HCC will increase as long-term survival with patients’ own livers increases. However, few reports have described the occurrence of malignant tumors such as HCC, hepatoblastoma, and cholangiocarcinoma [1-3], and no guideline for their follow-up has been established that considers the possibility of developing malignant liver tumors. Here we report a case of a 34-year-old man who developed HCC in his own liver during the follow-up after the surgery of BA.
2. Case Report
The patient was a 34-year-old man, who was born at a gestational age of 39 weeks with a birth weight of 2,926 g. He underwent a portal jejunostomy with a diagnosis of BA (IIIb1ν) on the 94th day after birth in our hospital. Thereafter, his course was generally smooth except for developing cholangitis at 3 months and 13 years after the surgery. CT at the age of 32 revealed bile stones in RY leg, liver right lobe atrophy, and left lobe hypertrophy, but not neoplastic lesion. Collateral circulation was also found, but endoscopy indicated that it required no treatment.
He visited our outpatient at the age of 34 with a chief complaint of fever and was admitted with a diagnosis of cholangitis. Contrast-enhanced CT imaging at the time of admission revealed a mass lesion with unclear borders in the right lobe of the liver and portal vein embolism that appeared to be continuous to it (Figure 1). HCC was suspected because portal vein embolism due to tumor was suspected. Measurements of tumor markers showed that AFP level was 1.3 ng/ml and PIVKA level was increased to 1,199 mAU/ml. Other blood test findings were T-Bil, 2.1 mg/dL; D-Bil, 1.2 mg/dL; AST, 293 IU/L; ALT, 207 IU/L; γ-GTP, 172 U/L; Alb, 2.7 mg/dL; APTT, 45.4 s; PT-INR, 1.73; FDP, 9.2 µg/mL; and D-dimmer, 3.5 µg/mL, which indicates the occurrence of mild clotting disorder. Child-Pugh classification was Grade B (score 9). Intrahepatic mass lesions could not be diagnosed in the contrast CT examination, so MRI was taken. MRI revealed a tumor that was not accompanied by ring enhancement in a T2-weighted image and gave low signals due to internal diffusion weighting at the same site as the above-mentioned tumor (Figure 2); thus, it was diagnosed as HCC together with an increase in tumor markers. Then, he was transferred to the department of gastroenterological medicine for treatment. The tumor embolism had already spread to the left branch of the portal vein, and resection and transplantation were considered contraindication. Proton therapy was started but was ineffective. He died 11 months after the detection of HCC.
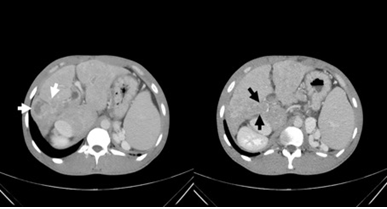
Figure 1: CT images of liver obtained on admission. Contrast-enhanced CT imaging demonstrated an enhanced mass lesion with unclear border in the right lobe of the liver (white arrow) and portal vein thrombosis (black arrow).
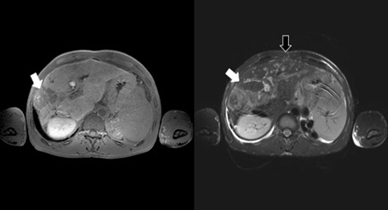
Figure 2: MR images obtained on admission. MRI revealed a tumor without ring enhancement in a T2-weighted image and gave low signals due to internal diffusion weighting at the same site as the tumor detected in CT imaging (white arrow). Multiple enhancing lesions with low signal central area in the left lobe indicated the hepatic micro abscesses due to cholangitis on a T2-weighted image (black arrow).
3. Discussion
Known complications after BA surgery include cholangitis, cholestatic cirrhosis, and bile stones, with few reports describing the occurrence of HCC in patients’ own liver [1-3]. The Japanese clinical practice guideline for biliary atresia [1] states that “the occurrence of hepatocellular carcinoma is 0.5–1.3% and the age of its onset ranges from 0.7 years old to adulthood”. A follow-up survey conducted by the Japan Biliary Atresia Society in 2016 found no reported case of HCC; however, such cases could have been treated as “long-term complications and others”. Okuyama et al. [5] reported the cases of HCC occurrence after BA surgery in 1965, and thereafter a total of 25 cases have been reported from Japan and overseas [5-21], of which 10 were from Japan [6-11, 18, 21].
Review of 26 cases including this case indicated that HCC had developed in the patients younger than 20 years old in 20 cases. Of these, 15 cases were the occurrence in the patients younger than 10 years old; thus, the occurrence in young people was noticeable (Table 1). Of these 20 cases of the occurrence in younger ages, all 17 with the description of BA history had failed to obtain reduced jaundice after hepatic portal jejunostomy. Even when reduced jaundice was obtained, the patients followed the course of repeated cholangitis or requiring reoperation. On the other hand, all the 3 cases including this case where HCC development was in their 30’s showed relatively good prognosis with reduced jaundice obtained in all of them [18, 21]. Taken together, these results suggest that early progression of cholestatic cirrhosis in BA due to poor reduction of jaundice and recurrent cholangitis is a risk factor of young onset of HCC. On the other hand, even in long-term survival cases with good reduction of jaundice, it is likely that the risk of HCC development increases as cholestatic cirrhosis progresses slowly over a long period of time. The cases like this case is expected to increase in the future. The small number of the reports of HCC cases is likely to be associated with the decrease in the number of patients who are living with their own liver under uncontrolled states due to the spread of liver transplantation. Regarding sex, 15 out of 26 cases were male. Considering that BA is more common among women, men may be more prone to develop HCC.
Primary biliary cirrhosis (PBC) is known for the occurrence of HCC on the basis of biliary cirrhosis [22-24]. The rate of complication with HCC for PBC is around 5%, which is lower than that of viral hepatitis [22-24]. Risk factors for the occurrence of HCC in PBC are considered to be being male, older age, history of blood transfusion, and the progression of histological stage [22-24]. Considering that HCC in BA occurs more frequently in men and that the progression and duration of having biliary cirrhosis are risk factors, multiple risk factors are common for the occurrence of HCC in BA and PBC. Therefore, cholestasis may also underlie the mechanism of HCC development in BA.
As for the opportunity of detecting HCC, only 8 cases out of 25 described were detected during regular follow-up of BA. Eight cases were detected accidentally during the scrutiny of jaundice and ascites as well as the examination for transplantation. In 9 cases, HCC was first detected at the time of necropsy or in transplanted livers. Therefore, early detection of HCC was considered to be a future challenge in the long-term follow-up of BA (Table 1). In BA, fibrosis of own liver parenchyma has progressed due to cholestasis, making it difficult to find tumors by ultrasonography alone. Abdominal ultrasonography only visualized the lesion as the one with unclear boundary in this case. Contrast-enhanced CT did not lead to diagnosis, either. And, MRI findings finally led to the diagnosis of HCC. Some reports recommend regular follow-up by MRI to ensure the accuracy and to avoid radiation exposure [13, 14, 16, 20]. These facts led us to consider it necessary to accumulate more data on the selection of subject cases (states) and the frequency of performing MRI.
Table 1: Clinical feature of patients with BA and HCC. CT - Computed tomography; MRI – Magnetic resonance imaging; US – Ultrasonography LT; Liver transplantation.
The course of this case and literature reviewing indicate that follow-up should be done on the assumption that patients’ own livers are in the states of cholestatic cirrhosis after the BA surgery even when jaundice is reduced and that they are at the risk of developing HCC. In addition, literature reviewing demonstrated that HCC can occur while young in the cases with poor reduction of jaundice and repeated cholangitis. We consider that long-term management considering the risk of carcinogenesis in cholestatic cirrhosis is required even for long-lived patients with their own livers, and that consulting with and transferring to adult gastroenterologists are also important. As experienced in this case, some HCCs can be difficult to find by abdominal ultrasonography or contrast-enhanced CT. In addition, it should be noted that AFP levels do not increase in some cases of HCC.
Ethics Approval and Consent to Participate
Not applicable
Consent for Publication
Written informed consent was obtained from the patient for publications of this case reports and any accompanying images.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Conflicts of Interest
The authors declare no conflicts of interest in association with the present study.
Authors' Contributions
Yasunari Tanaka, Hajime Takayasu, Kazunori Ishige and Kuniaki Fukuda conceived, coordinated and designed of study. Took care of the patient; Yasunari Tanaka and Toko Shinkai took care of the patient in the emergency service; Kouji Masumoto: helped to draft the manuscript; All authors read and approved the final manuscript.
Acknowledgements
We would like to thank Mr. Brian Quinn for editing the English language of this manuscript.
References
- Japanese Biliary Atresia Society. Japanese clinical practice guidelines for biliary atresia. Tokyo (2018): 83.
- Amir AZ, Sharma A, Cutz E, et al. Hepatoblastoma in Explanted Livers of Patients with Biliary Atresia. J Pediatr Gastroenterol Nutr 63 (2016): 188-194.
- Fukuda A, Sakamoto S, Kanazawa H, et al. Incidentally detected cholangiocarcinoma in an explanted liver with biliary atresia after Kasai operation. Pediatr Transplant 17 (2013): E62-E66.
- Parolini F, Boroni G, Milianti S, et al. Biliary atresia: 20-40-year follow-up with native liver in an Italian centre. J Pediatr Surg 54 (2019): 1440-1444.
- Okuyama K. Primary liver cell carcinoma associated with biliary cirrhosis due to congenital bile duct atresia: First report of a case. J Pediatr 67 (1965): 89-93.
- Tamura M, Hata Y, Sasaki F, et al. A case report with biliary atresia associated with hepatocellular carcinoma: J Jpn Soc Pediatr Surg 29 (1993): 131-135.
- Kawarasaki H, Iwanaka T, Tsuchida Y, et al. Partial liver transplantation from a living donor: experimental research and clinical experience. J Pediatr Surg 29 (1994): 518-522.
- Kohno M, Kitatani H, Wada H, et al. Hepatocellular carcinoma complicating biliary cirrhosis caused by biliary atresia: Report of a case. J Pediatr Surg 30 (1995): 1713-1716.
- Tatekawa Y, Asonuma K, Uemoto S, et al. Liver transplantation for biliary atresia associated with malignant hepatic tumors. J Pediatr Surg 36 (2001): 436-439.
- Azuhata T, Koshinaga T, Tanaka M, et al. Hepatocellular carcinoma complicating biliary atresia caused by HCV infection: a case report. J Jpn Soc Pediatr Surg 39 (2003): 765-770.
- Hori T, Michio K, Ikebukuro K, et al. Recurrence of hepatocellular carcinoma 5 years after liver transplantation in a child with biliary atresia and hepatocellular carcinoma. Jpn J Pediatr Hemator/Oncol. 41 (2004): 854-858.
- Brunati A, Feruzi Z, Sokal E, et al. Early occurrence of hepatocellular carcinoma in biliary atresia treated by liver transplantation. Pediatr Transplant 11 (2007): 117-119.
- Hol L, van den Bos IC, Hussain SM, et al. Hepatocellular carcinoma complicating biliary atresia after Kasai portoenterostomy. Eur J Gastroenterol Hepatol 20 (2008): 227-231.
- Iida T, Zendejas IR, Kayler LK, et al. Hepatocellular carcinoma in a 10-month-old biliary atresia child. Pediatr Transplant 13 (2009): 1048-1049.
- Had?i? N, Quaglia A, Portmann B, et al. Hepatocellular carcinoma in biliary atresia: King's College Hospital experience. J Pediatr 159 (2011): 617-622.
- Aggarwal S, Vadada D, Sharma V. A rare complication in an adult patient after Kasai portoenterostomy for biliary atresia. Arab J Gastroenterol 13 (2012): 148-149.
- Kim JM, Lee SK, Kwon CH, et al. Hepatocellular carcinoma in an infant with biliary atresia younger than 1 year. J Pediatr Surg 47 (2012): 819-821.
- Oyama T, Noda T, Yagi T?et al. A case report of biliary atresia associated with hepatocellular carcinoma treated by orthotopic liver transplantation. J Jpn Soc Pediatr Surg 48 (2012): 514.
- Zen Y, Vara R, Portmann B, et al. Childhood hepatocellular carcinoma: a clinicopathological study of 12 cases with special reference to EpCAM. Histopathology 64 (2014): 671-682.
- Hirzel AC, Madrazo B, Rojas CP. Two Rare Cases of Hepatocellular Carcinoma after Kasai Procedure for Biliary Atresia: A Recommendation for Close Follow-Up. Case Rep Pathol 2015 (2015): 982679.
- Arai Y, Kubota M, Kobayashi T, et al. A case of Synchronous combined cancer with Intrahepatic cholangiocarcinoma and hepatocellar carcinoma 39 years after operation for biliary atresia. J Soc Jpn Pedeiatr Surg 52 (2016): 1303-1308.
- Suzuki A, Lymp J, Donlinger J, et al. Clinical predictors for hepatocellular carcinoma in patients with primary biliary cirrhosis. Clin Gastroenterol Hepatol 5 (2007): 259-264.
- Rong G, Wang H, Bowlus CL, et al. Incidence and risk factors for hepatocellular carcinoma in primary biliary cirrhosis. Clin Rev Allergy Immunol 48 (2015): 132-141.
- Shibuya A, Tanaka K, Miyakawa H, et al. Hepatocellular carcinoma and survival in patients with primary biliary cirrhosis. Hepatology 35 (2002): 1172-1178.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
