In The Era of Precision Medicine- Where Does the Diagnosis “Hypertensive Kidney Disease” Fit?
Article Information
Sadichhya Lohani MBBS1*, Mohamed G Atta MD, MPH2
1Renal-Electrolyte and Hypertension Division, University of Pennsylvania, Pennsylvania, United States
2Division of Nephrology, The Johns Hopkins University School of Medicine, Maryland, United States
*Corresponding Author: Dr. Sadichhya Lohani, Renal-Electrolyte and Hypertension Division, University of Pennsylvania, Pennsylvania, United States
Received: 20 January 2020; Accepted: 31 January 2020; Published: 02 March 2020
Citation: Sadichhya Lohani, Mohamed G. Atta. In The Era of Precision Medicine- Where Does the Diagnosis “Hypertensive Kidney Disease” Fit?. Archives of Clinical and Medical Case Reports 4 (2020): 164-179.
View / Download Pdf Share at FacebookAbstract
Hypertension has been labeled as a cause of non-diabetic chronic kidney disease for decades. In today’s era of precision medicine, labeling benign hypertension as the initial etiology for chronic kidney disease might not be accurate. We discuss an example of a 55-year-old African American patient with pathologic findings of nephrosclerosis without history of hypertension. The pathologic findings of nephrosclerosis can be as a result of obesity, aging, genetic susceptibility and variety of other previously undiagnosed primary renal disease, and hence shouldn’t automatically mark the diagnosis of hypertensive nephrosclerosis. Multiple studies to date have failed to prove or refute causality between benign hypertension and non-diabetic chronic kidney disease. Genome sequencing of non-diabetic kidney diseases have identified mutations in Apolipoprotein L1 gene, MYH9 gene and uromodulin gene as a risk factors for chronic kidney disease. There is no question that uncontrolled hypertension leads to progression of kidney disease, and hence blood pressure should be controlled adequately. The relationship between hypertension and kidney dysfunction should be viewed as an association and broad differential diagnoses should be pursued.
Keywords
Hypertensive nephrosclerosis; Benign hypertension; Non-diabetic chronic kidney disease; Genetic diseases of kidney
Article Details
1. Introduction
Hypertensive nephrosclerosis has been described as an important public health problem and a cause for End Stage Kidney disease (ESKD) for decades [1-4]. About 30 million American adults have chronic kidney disease (CKD) and roughly 32% of American adults with CKD have hypertension [5]. Approximately 6% of patients with essential hypertension have co-existing chronic kidney disease and are at risk for progression to ESKD [6], mechanism of acute kidney injury (AKI) as evidence of end organ damage from malignant hypertension is well understood and recurrent episodes of acute kidney injury with episodes of hypertensive emergency leads to progression of CKD and ESKD. Hypertension also perpetuates the progression of CKD, and hence the importance of controlling the blood pressure to appropriate goal. There is still debate, however, about whether benign essential hypertension causes CKD, or if underlying kidney disease is the cause of hypertension in patients who are labeled as hypertensive nephrosclerosis [7-9]. Apolipoprotein L1 (APOL1) genotype variants and uromodulin gene variants, among other risk factors, are now being increasingly linked to both hypertension and CKD. This should make us re-think and shift our paradigm from hypertension as a cause for CKD to rather a reflection of underlying primary kidney disease.
2. Case Discussion
A 55-year-old African American man presented to the hospital with intermittent nausea and fatigue. He was noted to have a creatinine peak of 7.1 mg/dl from his baseline of 2.6-3.2 mg/dL. He had a history chronic kidney disease of unknown etiology for 6 years with serum creatinine of 1.5 mg/dl 4 years prior to presentation. His blood pressure (BP) on presentation was 133/75 mmHg and remained controlled during the seven-day hospital stay with average blood pressure ranging from 113-125/ 60-75 mmHg with occasional systolic readings of 140-150 mmHg. He didn’t have access to regular health care. His body mass index (BMI) was 27 kg/m2. He didn’t have a history of hypertension, diabetes mellitus, coronary artery disease, nephrolithiasis, or pyelonephritis. He had a history of untreated hepatitis C with viral load of 1,115 copies/ml diagnosed a year prior to this presentation. A year prior to this admission, his blood pressure was 125/88 mmHg. There was documentation of one isolated systolic blood pressure of 140 mmHg a year prior. Even prior to the diagnosis of CKD 6 years ago, he didn’t have history of high blood pressure. He was never on any antihypertensive medications in the past. His urine protein: creatinine ratio a year prior to the presentation was 460 mg/g. Home medications included calcium and magnesium supplements for cramps. He had history of chronic back pain. He had remote use of intermittent non-steroidal anti-inflammatory agents (NSAIDS). He was born at term and didn’t have any complications at birth or major illnesses during childhood. He didn’t have a family history of kidney disease or hypertension. He didn’t have history of smoking, alcohol or recreational drugs. He didn’t have history of blood transfusion. He worked as a driver and didn’t have occupational exposure to toxic chemicals or heavy metals.
Upon further evaluation, on this admission his 24-hour urine protein was 6.7 g. A kidney ultrasound showed a right kidney measuring 10.7 cm and left kidney measuring 11 cm without hydronephrosis. Urine sediment showed few white cells, non-dysmorphic red cells and many waxy casts per high power field. Complement levels were low, with C3 49 mg/dL (79-152) and C4 13 mg/dL (12-42). Free light chains were normal. Uric acid level was 9.0 mg/dL (3.5-7.2 mg/dL). Serum protein electrophoresis didn’t reveal monoclonal protein. HIV was negative on ELISA. ANA, rheumatoid factor, cryoglobulins were negative. Lactate dehydrogenase was 324 U/L (118-273) and haptoglobin of 37 mg/dL (36-196mg/dL).
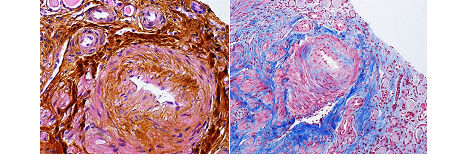
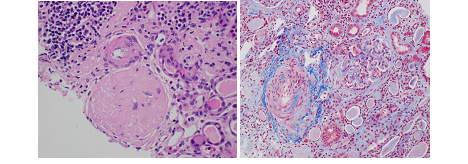
In addition to worsening renal function, the proteinuria had worsened from 460mg/g a year prior to 6.7 g on admission. With the initial differential diagnosis of hepatitis C related kidney disease considering proteinuria, hypocomplementemia and history of untreated hepatitis C infection, a kidney biopsy was performed. The biopsy specimen consisted of three cores containing approximately 80% renal cortex and 20% medulla. The biopsy showed 9 glomeruli of which 55% were globally sclerosed. Non-obsolescent glomeruli have prominent periglomerular fibrosis and variable glomerular capillary wall remodeling/reduplication with ischemic type changes. Tubular atrophy and interstitial fibrotic expansion involve approximately 60% of the cortex involves approximately 60% of the cortex. Of the 3 interlobular arteries sampled, severe fibro-intimal thickening, fibroelastosis, medial hypertrophy was noted, arterioles with focal “onion skinning changes with severe luminal constriction suggestive of severe arteriosclerosis and arteriolosclerosis were present. The immunofluorescence (IF) pattern was non-specific not favoring hepatitis C mediated process. Electron microscopy (EM) revealed segmental podocyte foot process fusion (approximately 50%) consistent with secondary focal segmental glomerulosclerosis or FSGS; the glomerular basement membranes were thickened with segmental ischemic type wrinkling. No immune complex type deposits were identified. (Figure 1 and 2). These biopsy findings were reported to be consistent with vascular mediated glomerular injury (glomerular hypoperfusion) secondary to severe chronic renovascular sclerosis.
Figure 1: Renal biopsy showing arteriosclerosis on light microscopy (Trichrome and Silver stain). Light microscopy with trichrome (left) and silver stain (right). There is severe fibrointimal thickening, fibroelastosis and medial hypertrophy. Arterioles show focal 'onion-skinning' changes with severe luminal constriction. Significant arteriolar hyalinosis is not appreciated.
Figure 2: Renal biopsy showing global glomerulosclerosis and periglomerular fibrosis on light microscopy (H&E and Trichrome). Light microscopy with H&E(left) and trichrome (right) stain. Globally sclerosed glomeruli seen. Non-obsolescent glomerulus has prominent periglomerular fibrosis. Mesangial matrix is mildly and segmentally expanded with no significant increase in cellularity. There are no crescents, necrosis or thrombi identified.
Interestingly, the patient didn’t have history of uncontrolled hypertension which was supported by his blood pressure trends in the hospital and limited blood pressure readings on ambulatory settings. Because ambulatory readings were not possible to obtain due to patient’s lack of routine outpatient care, periodic masked hypertension outside of the hospital would be difficult to obtain. Hence, in addition to close monitoring of his blood pressure in the hospital, work up for secondary causes of hypertension for episodic masked hypertension were pursued. He only had mildly elevated plasma metanephrines at 66 pg/mL (reference <57) with normal normetanephrine level at 142 pg/ml (reference <148). 24-hour urine nor-metanephrine was 351 mcg (reference-122-676 mcg/24hr) and total epinephrine plus norepinephrine was 7 (26-121 mcg/24 h). These findings wouldn’t support diagnosis of pheochromocytoma. His clinical presentation was also not suggestive of pheochromocytoma.
The diagnosis of hypertensive nephrosclerosis has been given to many patients with CKD based on the described pathologic findings. The findings of severe arteriosclerosis and arteriolosclerosis in this patient without history of uncontrolled hypertension should make us think if we have been labeling patients incorrectly as hypertensive nephrosclerosis. Features of nephrosclerosis in this patient could have been related to primary renal disease from untreated hepatitis C or underling genetic predisposition from Apolipoprotein (APOL1) mutation (differential diagnoses in table 1). The clinical course of patient included improvement in clinical symptoms and was discharged with follow up with nephrologist and plans to initiate renal replacement therapy on non-urgent basis.
|
Disease |
Supporting |
Against |
|
Hepatitis C related chronic membranoproliferative glomerulonephritis with chronicity and secondary FSGS |
Untreated hepatitis C; Low complements; Proteinuria; Global and focal segmental sclerosis due to chronicity |
No immune complex deposits on electron microscopy; Non-specific immunofluorescence |
|
APOL1 mediated global and focal segmental sclerosis |
Biopsy findings of global sclerosis on light and foot process effacement on electron microscopy, absence of immune complex on electron microscopy |
Hypocomplementemia Absence of family history |
|
Chronic thrombotic microangiopathy related to malignant hypertension |
Severe fibro-intimal thickening, fibroelastosis and medial hypertrophy of interlobular arteries; ‘Onion-skinning' changes with severe luminal constriction of arterioles; Glomerular capillary wall remodeling/reduplication with ischemic-type changes |
LDH is only mildly elevated Haptoglobin is normal Blood pressures are not severely elevated |
|
Obesity related kidney disease |
Focal segmental glomerulosclerosis; Thickened glomerular basement membrane; Arteriosclerosis and arteriolosclerosis Body mass index: 27; nephrotic range proteinuria |
Absence of glomerulomegaly Absence of morbid obesity |
Table 1: Differential diagnosis of the Nephrosclerosis.
3. Discussion of Literature Review
3.1 The burden of hypertension
Hypertension has been known for many decades to be a major risk factor for myocardial infarction, heart failure, stroke, peripheral artery disease, atrial fibrillation and chronic kidney disease, and has been referred to as a “silent killer” [10]. The burden of hypertension in cardiovascular disease is well established. Hypertension treatment has been shown to decrease cardiovascular risk as demonstrated in the Veteran’s Affairs Cooperative trials of the late 1960s to 1970s [11,12]. However, hypertension is an aging disorder, and in the United States, among the estimated 85.7 million adults with hypertension (defined as blood pressure (BP) greater than or equal to 140/90 mm Hg) <10% of persons aged <40 years compared to two-thirds of persons aged ≥ 60 years have hypertension [10,13]. According to United Stated Renal Data System (USRDS) annual report prevalence of CKD in patients with hypertension was reported as 31% [14]. Despite the widespread reporting of hypertension as a cause of CKD, the mechanism of non-malignant hypertension as a risk factor of chronic kidney disease is a subject of an interesting debate.
3.2 Definition and history of hypertensive nephrosclerosis
Hypertensive nephrosclerosis has been defined in the literature as a non-diabetic chronic kidney disease associated with chronic hypertension, with or without proteinuria, and a pathologic picture characterized by arteriosclerosis, arteriolosclerosis, glomerulosclerosis, and interstitial fibrosis without immune deposits [1, 10]. The term nephrosclerosis was initially derived from the Greek word “nephros, kidney; sklerosis, hardening,” resulting from dense collagenous tissue replacing normal kidney parenchyma attributed to hypertension [11]. The history of hypertensive nephrosclerosis first started in 1873 when Dr. Stewart described the pathologic findings in a fibrotic kidney which was previously described as “the morbid changes in the arteriole and capillaries in the primary and essential condition of the morbid state called chronic Bright’s disease with contracting kidneys” [12]. However, it remains to date a topic of debate whether the non-malignant hypertension is indeed the culprit for this scarring in these kidneys labeled as hypertensive nephrosclerosis.
3.3 How is nephrosclerosis described?
Hypertension is described as a cause for renal vascular injury, including ischemia and hypertrophy of glomeruli, vessels, and tubules [1, 13]. Arteriosclerosis and arteriolosclerosis are characterized by muscular hyperplasia leading to narrowing of arteriolar lumen, intimal fibrosis, arteriolar hyalinosis that appears as focal lesions with homogenous eosinophilic acellular material that stains brightly on periodic acid-Schiff staining [1, 11]. The interlobar arteries have fibroelastic thickening and medial scarring [11]. Other pathologic findings are global glomerulosclerosis predominantly obsolescence characterized by collapse of the glomerular tuft and intracapsular fibrosis, segmental glomerulosclerosis, tubulointerstitial changes, tubular atrophy, inflammation and fibrosis [1, 14, 15]. These pathologic findings summarized as nephrosclerosis have been attributed in literature and in practice to preceding hypertension induced preglomerular microvascular injury, atherosclerotic narrowing of preglomerular arteries and arterioles leading to reduced glomerular perfusion [11, 16].
3.4 Is hypertension a cause for CKD or mere association?
Since the 1800s, hypertension has remained the second most common etiology for ESKD [10]. Systolic hypertension has been reported as a powerful predictor of the development of ESKD [17, 18]. Hypertensive urgency as a cause for acute kidney injury with fibrinoid necrosis in the arterioles from the shear stress on the vascular wall is understandable, but the pathogenesis of hypertensive nephrosclerosis from essential hypertension still remains unclear [10]. MRFIT trial had shown a strong graded association of systolic and diastolic blood pressures with development of ESKD irrespective of other demographic variables, diabetes, myocardial infarction, smoking and dyslipidemia [17]. However, baseline kidney function was not assessed in the majority of this cohort making it unclear if renal dysfunction was already present in patients who developed ESKD later [17]. In a subsequent study, Iseki et al. adjusted the baseline serum creatinine, proteinuria and hematuria to demonstrate that high blood pressure alone was, in fact, not a risk factor for ESKD [19]. In the African American Study of Kidney Disease (AASK) Study, kidney biopsies were performed in 39 patients, with mean blood pressures were 109 +/- 15 and GFR 52+/-13 ml/min which showed findings of arteriosclerosis/arteriolosclerosis, glomerulosclerosis, basement membrane thickening, global glomerulosclerosis, tubular atrophy and interstitial fibrosis [13]. Based on these pathologic findings with correlation to their systolic blood pressures, the authors concluded that the non-diabetic hypertensive African American patient have renal vascular changes supportive of hypertensive nephrosclerosis [13]. However, these biopsies and blood pressure correlation were cross-sectional findings with no prior baseline renal function or the trends in blood pressures to show if the renal findings preceded or followed the rise in blood pressure. All the epidemiologic and observational studies demonstrate that hypertension and CKD are closely linked, but we cannot prove causality and make a statement that benign hypertension causes CKD.
The argument against causality linking hypertension to CKD is not new. In 1993, Perneger stated that progression to ESKD is rare in hypertension related kidney disease and other factors likely play a role. He had noted that each year, out of the approximately 1.8 million middle-aged Americans who develop hypertension, only 140,000 develop hypertension-related kidney dysfunction, and only 5,300 develop hypertension-related ESKD [20]. This raises an important argument that all hypertensive patients do not develop chronic kidney disease and subsequent ESKD. Another important question this brings to discussion-does treatment of hypertension influence the development of chronic kidney disease and subsequent ESKD? If benign hypertension causes CKD, the treatment and control of blood pressure should prove such as well. Hsu in 2001 conducted a metanalysis of 26,521 individuals with non-malignant hypertension from 10 randomized control trials with treatment consisting of diuretics and adrenergic blockers for hypertension [7]. This study demonstrated that there is no difference in relative risk of developing kidney dysfunction among patients randomized to antihypertensive therapy with relative risk of 0.95 (95% confidence interval of 0.58-1.55, p value of 0.48) [7]. This meta-analysis was repeated including secondary analysis, and still didn’t show reduction in incidence of kidney dysfunction in the treatment versus control group, with a relative risk of 0.97 (95% CI:0.78-1.21, p=0.77). (7) In another cohort of 2,125 mild to moderate hypertensive men, in-treatment diastolic pressure was not independently associated with final serum creatinine [21]. The development of kidney dysfunction in patients with non-malignant hypertension irrespective of treatment suggests that renal dysfunction is most likely a result of an intrinsic kidney disease. Even though treatment of mild HTN does not seem to affect renal outcomes unlike effect on cardiovascular outcomes, the treatment of hypertension with blockers of the renin angiotensin aldosterone blockade may have an impact on the kidney disease especially with proteinuria and hence should be treated.
Hypertension is often implicated as the cause of most non-diabetic CKD that doesn’t have other obvious etiology. The pathogenesis for development of kidney injury has been poorly described for benign hypertension but is better explained when hypertension and CKD are considered as an association taking in to account genetic predisposition, racial differences in susceptibility to hypertension, and kidney damage [1, 4]. Several studies have suggested that reduced nephron number and nephron hypertrophy play a role in the development of hypertension and the progression of renal disease [1, 22-25]. In an autopsy study by Hughson et al, hypertensive subjects had larger glomeruli compared to non-hypertensive subjects, independent of race or gender [16]. African American males had significantly larger glomeruli than white males [16]. As postulated by Hughson, the glomerular hypertrophy and hypertension could have developmental associations with glomerulosclerosis and arterial intimal thickening which appears to be more common in African American population. In the African American Study of Kidney Disease (AASK) Study, African Americans had more severe arteriosclerosis, glomerulosclerosis and more frequent solidification of glomeruli compared to Whites irrespective of difference in blood pressures due to race in the biopsy series of patients labeled as hypertensive nephrosclerosis [16, 26, 27]. These findings do not support or refute the causality between the findings of nephrosclerosis and hypertension but rather suggests an association.
A study of 94 patients treated for benign hypertension showed despite good control of diastolic blood pressure (less than or equal to 90 mm Hg), African American patients were twice as likely as Caucasian patients to have elevations in serum creatinine (23 percent vs. 11 percent) [28]. Lipkowitz also described that the progressive decline in renal function was unlikely caused by essential hypertension as the results of decline in renal function were consistent when comparing hypertensive AASK cases to controls with or without high BP and lower blood pressure goal didn’t slow the progression of renal disease in African American population [29]. In numerous studies like these , ESKD attributed to hypertension has been more strongly associated in African Americans than white population [18]. This should bring to attention that there should be another risk factor independent from blood pressure that is causing renal dysfunction in African American population that predisposes them to both with CKD and hypertension.
3.5 Genetics behind the hypertension and CKD
Studies showing a racial predisposition and non-significant effect of treatment on the development of kidney disease suggest that there may be another independent risk factor for CKD and hypertension. Now, we have increasing data on this racial predisposition of presumed hypertension attributed CKD and ESKD, with the identification of the region on chromosome 22q that contains the genes- non-muscle myosin heavy chain type 2 (MYH9) and apolipoprotein-L1 (APOL1) [29, 30]. The association of non-diabetic kidney disease including FSGS and hypertension in African American population with APOL1 variants has provided a greater understanding about this racial predisposition as well [16].
APOL1 gene variants are known to be associated with non-diabetic chronic kidney disease and ESKD in African Americans while MYH9 risk alleles with individuals of European ancestry [29]. The association has been reported at a higher rate for variants in APOL1 gene and African American ancestry than with white patients [31]. The APOL1 two-risk variants were present in 10% of patients with hypertension-attributed CKD, 6% of their first-degree relatives and 8.6% of controls [31]. These variants of APOL1 (termed G1 and G2) had evolved to protect from a species of trypanosomal infection in African population and now has been carried worldwide in descendants of the population from the geographic region [32]. G1 and/or G2 APOL1 risk variants were present in more than 30% of African American chromosomes [33]. Approximately 12% of HIV-negative and 50% of HIV-positive African American APOL1 G1/G2 homozygotes or compound heterozygotes may be susceptible to kidney disease [30]. Apolipoproteins are structurally and functionally similar to proteins in the BCl2 family which play a role in apoptosis and hence the possibility that it has role in the apoptosis in the kidney resulting in glomerulosclerosis [30, 34]. The G1 and G2 APOL1 variants have dysunctional particle which is potential etiology for inflammation in the renal vascular endothelisum resulting in arteriolar nephrosclerosis as a pathogenesis of CKD [30].
African American subjects with one APOL1 risk allele vs zero risk allele have odds ratio (OR) of 1.26 and with two risk alleles vs zero risk allenes have OR of 7.3 for ESKD labeled as hypertension associated with relation to susceptibility for glomerulosclerosis [16, 30, 33]. It has been postulated that APOL1 risk alleles may be associated with glomerular enlargement [35]. Glomerular number (Nglom) and mean glomerular volume (Vglom) were measured in kidneys of African-American and non–African-American adults without renal disease, undergoing autopsies in Jackson, Mississippi and their APOL1 risk alleles were genotyped [35]. The proportions of African Americans with zero, one, and two APOL1 risk alleles were 38%, 43%, and 19%, respectively; 38% of African Americans had G1 allele variants and 31% of African Americans had G2 allele variants [35]. Only APOL1-positive African Americans had significant reductions in Nglom but with increase in volume with increasing age [35]. AASK cohort had findings of focal global glomerulosclerosis, arteriolar nephrosclerosis and interstitial scarring in the renal biopsies which is this disease-spectrum based on APOL1 association [13]. This would suggest that the pathologic findings in many of the patients labeled as hypertensive nephrosclerosis may have APOL1 risk variants. This has been demonstrated in the prospective clinical outcome study of ESKD by Foster in 2013 in which the presence of two risk alleles conferred a 1.49-fold increased risk of CKD (95% CI=1.02 to 2.17) and a 1.88-fold increased risk of ESKD (95% CI=1.20 to 2.93) compared with zero or one risk allele [36]. Many studies have conclusively confirmed the causal link beteween APOL1 gene variants and CKD [4, 30, 33, 36]. This also explains the racial differences in the susceptibility to renal damage from elevated blood pressures and increased risk for ESKD attributed to hypertension in African Americans.
In addition to APOL1 gene variants associated with CKD, recent data have confirmed other missed genetic diagnoses with exome sequencing of CKD cohorts [37]. We have enough evidence that requires reconsideration and further exploration of other genetic risk factors and causes for development of CKD and progression to ESKD in patients with benign hypertension. Recently another entity known as serum uromodulin, has been described as independently associated with progression of CKD and ESKD [38]. Mutations in the uromodulin (UMOD) gene, which encodes uromodulin (Tamm-Horsfall protein) have been identified as a cause of hereditary autosomal-dominant tubulointerstitial diseases [38-40]. The variants in UMOD gene have also been recognized as the risk factors for CKD and hypertension [38, 41-43].
The relationship between hypertension and kidney dysfunction needs to be labeled as an association until causality is conclusively established. Nephrosclerosis on kidney biopsy is mostly driven by aging but also by obesity, and potentially genetic susceptibility. Hypertension is potentially a consequence or a simultaneous manifestation of an undiagnosed kidney parenchymal disease or renal vascular disease [11]. We know that blood pressure reduction in chronic kidney disease decreases risk of progression, regardless of the etiology of chronic kidney disease [44]. Uncontrolled hypertension potentiates the injury to kidney parenchyma and vessels and should be adequately treated, but we would like to suggest that mild to moderate elevations in high blood pressure is likely not the initial insult for the kidney dysfunction.
|
Study |
Type /Study population |
Results |
Conclusion |
|
Kasiske. Kidney International. 1987 [45] |
Autopsy study, N=114 Group I: mild systemic atherosclerosis, N=57; Group II: moderate to severe atherosclerosis, N=57 |
Incidence of sclerotic glomeruli: Group I: 8.3 +/- 7.0%; Group II: 15.4 +/- 16.3% (P ± 0.01). Relative intrarenal arterial wall area increased in group II (60+/- 12%) vs group I (55 +/-11%, P=0.05). Difference in intrarenal arterial wall thickening between Group I vs Group 2: -0.32 (p=<0.01) with hypertension |
Age and intrarenal arterial disease were independently correlated to glomerulosclerosis. Hypertension or use of antihypertensive was not independently associated with glomerulosclerosis after age and intrarenal vascular disease was accounted for. |
|
Rostand et al. NEJM. 1989. [28] |
Prospective, cohort study, N=94, Serum Creatinine <1.5 mg/dL Follow up: 34 months. |
16% of the 61 patients with good control of blood pressure, the serum creatinine concentration rose 0.67 +/- 0.38 g/dL Rise in creatinine: 23% in Black and 11% in White with diastolic BP <90 |
Despite good blood pressure control, some patients especially black continued to have progressive renal dysfunction suggesting hypertension is not the sole cause for the kidney disease. |
|
Walker et al. JAMA. 1992. [47] |
Retrospective cohort N=5524 (463 black, 5061 non-black), men Age: 35-57 limited evidence of end-organ damage |
Mean creatinine slope: Black men: 0.0090 +/- 0.0013 dL/mg/y) vs white men:(+0.0018 +/- 0.0004 dL/mg/y) (P=0.001) Sustained diastolic BP>95 decline at -0.0013 ± 0.0009 dL/mg/y (P=0.007 for difference) |
Decline in renal function was greater among high systolic and diastolic BP and with older age. Effective blood pressure control was associated with stable or improving renal function in white men but not in blacks. |
|
Fogo et al. Kidney International. 1997 [13] |
Renal biopsy on eligible AASK cohort N=88 (39 biopsies) African American, Age:18-70; Diastolic BP >95 and GFR 25-70ml/min/ 1.73 m2 |
MAP: 109 +/- 15 mm Hg and GFR 51.7 +/- 13.6 ml/min/1.73 m2 38/ 39 biopsies: arteriosclerosis (1.5 +/- 0.9) and/or arteriolosclerosis (1.5 +/- 0.8), moderate interstitial fibrosis (1.3 +/- 0.9) on 0 to 3+ scale. Global glomerulosclerosis (43 +/- 26% of glomeruli) The degree of findings correlated with systolic blood pressure with P=0.0174) |
Renal vascular lesions in non-diabetic chronic kidney disease without marked proteinuria correlated with blood pressure. This was a cross sectional study and baseline BP, renal function was not studied and findings more likely demonstrates associations rather than causality. |
|
Hsu. Journal of human hypertension. 2001 [7] |
Meta-analysis of 10 Randomized controlled trials (RCT) N=26521 |
Relative risk of renal dysfunction in treatment of blood pressure vs control was 0.95 (CI: 0.58-1.55, P= 0.48) |
Among patients with non-malignant hypertension, treated patients did not have a lower risk of renal dysfunction. |
|
Foster et al. JASN. 2013. [36] |
Prospective observational study N=3067, African Americans, no baseline CKD in the Atherosclerosis Risk in Communities Study |
1.49-fold increased risk of CKD with 2 risk alleles for APOL1 (CI=1.02 to 2.17) and a 1.88-fold increased risk of ESKD (CI=1.20 to 2.93) compared to 0 or 1 risk allele; CKD: 2 risk alleles more likely to progress to ESKD than 0 or 1 risk allele (HR=2.22, CI=1.01 to 4.84) |
APOL1 risk alleles are associated with development of CKD and progression to ESKD. |
|
Hughson et al. Nephrol Dial Transplant. 2014 [16] |
Retrospective cohort with human autopsy tissues N=171 African Americans and 131 Caucasian Hypertension: African Americans:58.5%; whites: 35.8% (P=0.02). |
African American: increased volume of glomeruli V glom (P=0.01) and arterial intimal thickening (P ± 0.01) Cortical fibrosis and arterial intimal thickening were statistically correlated with age (P ± 0.001), hypertension (P ± 0.001), increased Vglom with hypertension (P ± 0.001) and BMI (P ± 0.001) for each race and gender, glomerulosclerosis. |
APOL1-positive African Americans without hypertension or cardiovascular deaths had reduced N glom with age. Glomerular hypertrophy was a feature of hypertensive nephropathy and preceded glomerulosclerosis. Race affected Vglom and arterial intimal thickening relating to the more frequent and severe hypertension among African Americans. |
|
Hoy WE et al, JASN. 2015. [35] |
Retrospective, autopsy study in Jackson, Mississippi in patients without renal disease N=159African Americans; 135 non–African Americans |
African Americans with APOL1 risk alleles: none (38%), 1 (43%), and 2(19%) African Americans with G1 allele: 38% and G2 allele: 31% APOL1-positive African Americans had significant reductions in number of glomeruli and increases in Vglom with age. |
The age-related decrease in glom in APOL1-positive people could not be readily attributed to hypertension or cardiovascular disease. |
|
Li Lv et al. Journal of translational medicine 2018 [38] |
Prospective study with 53.6 months median follow up N=2652. CKD patients from the Chinese Cohort Study of Chronic Kidney Disease (C-STRIDE) |
Hazard ratio for risk of ESKD for low level of uromodulin was 1.92 (95% CI 1.26-2.90) and 0.69 (95% CI 0.55-0.86) per every standard deviation increase despite adjusting for baseline eGFR. |
Lower levels of serum uromodulin were independently associated with higher risk of incident ESKD. This hints that there are newer biomarkers and genetic variants being identified that have associations with CKD. |
Table 2: Summary of studies.
4. Conclusion
We have enough evidence to support that the genetic variants are associated with pathologic findings on kidney biosies that drive renal dysfunction as well as hypertension. Future genetic studies are needed in the CKD population to identify genetic variants that are associated with these pathological findings, particularly in African Americans. More prospective large population studies with non-malignant hypertension taking into account baseline creatinine, proteinuria, genetic predisposition, and effect of antihypertensive treatment would be ideal to demonstrate the phenomenon of either association or causality between benign hypertension and CKD. Nephrosclerosis is a pathologic finding that requires investigation of potential causes rather than been spontaneously linked to non-malignant hypertension. We desire to advocate for precision medicine and banning labeling our non-diabetic CKD patients with benign hypertension as hypertensive nephrosclerosis.
Conflict of Interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
References
- Toto RB. Hypertensive nephrosclerosis in African Americans. Kidney international 64 (2003): 2331-2341.
- Whelton PK, Perneger TV, He J, et al. The role of blood pressure as a risk factor for renal disease: a review of the epidemiologic evidence. J Hum Hypertens 10 (1996): 683-689.
- Klag MJ, Whelton PK, Randall BL, et al. End-stage renal disease in African-American and white men. 16-year MRFIT findings. Jama 277 (1997): 1293-1298.
- McClellan W, Tuttle E, Issa A. Racial differences in the incidence of hypertensive end-stage renal disease (ESRD) are not entirely explained by differences in the prevalence of hypertension. American journal of kidney diseases : the official journal of the National Kidney Foundation 12 (1988): 285-290.
- Martin LD, Howell EE, Ziegelstein RC, et al. Hospitalist performance of cardiac hand-carried ultrasound after focused training. The American journal of medicine 120 (2007): 1000-1004.
- Shulman NB, Ford CE, Hall WD, et al. Prognostic value of serum creatinine and effect of treatment of hypertension on renal function. Results from the hypertension detection and follow-up program. The Hypertension Detection and Follow-up Program Cooperative Group. Hypertension 13 (1989): I80-I93.
- Hsu CY. Does treatment of non-malignant hypertension reduce the incidence of renal dysfunction? A meta-analysis of 10 randomised, controlled trials. Journal of human hypertension 15 (2001): 99-106.
- Kincaid-Smith P. Renal pathology in hypertension and the effects of treatment. Br J Clin Pharmacol 13 (1982): 107-115.
- Weisstuch JM, Dworkin LD. Does essential hypertension cause end-stage renal disease? Kidney Int Suppl 36 (1992): S33-S37.
- Luft FC. Hypertensive nephrosclerosis-a cause of end-stage renal disease? Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 15 (2000): 1515-1517.
- Freedman BI, Iskandar SS, Appel RG. The link between hypertension and nephrosclerosis. American journal of kidney diseases : the official journal of the National Kidney Foundation 25 (1995): 207-221.
- Stewart TG. Remarks on Chronic Bright's Disease, Particularly the Cirrhotic Form. Br Med J 2 (1873): 565-567.
- Fogo A, Breyer JA, Smith MC, et al. Accuracy of the diagnosis of hypertensive nephrosclerosis in African Americans: a report from the African American Study of Kidney Disease (AASK) Trial. AASK Pilot Study Investigators. Kidney international 51 (1997): 244-252.
- Glassock RJ, Rule AD. The implications of anatomical and functional changes of the aging kidney: with an emphasis on the glomeruli. Kidney international 82 (2012): 270-277.
- Rule AD, Cornell LD, Poggio ED. Senile nephrosclerosis--does it explain the decline in glomerular filtration rate with aging? Nephron Physiol 119 (2011): 6-11.
- Hughson MD, Puelles VG, Hoy WE, et al. Hypertension, glomerular hypertrophy and nephrosclerosis: the effect of race. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 29 (2014): 1399-1409.
- Klag MJ, Whelton PK, Randall BL, et al. Blood pressure and end-stage renal disease in men. The New England journal of medicine 334 (1996): 13-18.
- Perry HM, Miller JP, Fornoff JR, et al. Early predictors of 15-year end-stage renal disease in hypertensive patients. Hypertension (Dallas, Tex : 1979) 25 (1995): 587-594.
- Iseki K, Ikemiya Y, Fukiyama K. Risk factors of end-stage renal disease and serum creatinine in a community-based mass screening. Kidney international 51 (1997): 850-854.
- Perneger TV, Klag MJ, Feldman HI, et al. Projections of hypertension-related renal disease in middle-aged residents of the United States. Jama 269 (1993): 1272-1277.
- Madhavan S, Stockwell D, Cohen H, et al. Renal function during antihypertensive treatment. Lancet (London, England) 345 (1995): 749-751.
- Brenner BM, Chertow GM. Congenital oligonephropathy and the etiology of adult hypertension and progressive renal injury. American journal of kidney diseases : the official journal of the National Kidney Foundation 23 (1994): 171-175.
- Keller G, Zimmer G, Mall G, et al. Nephron number in patients with primary hypertension. N Engl J Med 348 (2003): 101-108.
- Rossing P, Tarnow L, Nielsen FS, et al. Low birth weight. A risk factor for development of diabetic nephropathy? Diabetes 44 (1995): 1405-1407.
- Spencer J, Wang Z, Hoy W. Low birth weight and reduced renal volume in Aboriginal children. American journal of kidney diseases : the official journal of the National Kidney Foundation 37 (2001): 915-920.
- Marcantoni C, Ma LJ, Federspiel C, et al. Hypertensive nephrosclerosis in African Americans versus Caucasians. Kidney international 62 (2002): 172-180.
- Marcantoni C, Fogo AB. A perspective on arterionephrosclerosis: from pathology to potential pathogenesis. J Nephrol 20 (2007): 518-524.
- Rostand SG, Brown G, Kirk KA, et al. Renal insufficiency in treated essential hypertension. N Engl J Med 320 (1989): 684-688.
- Lipkowitz MS, Freedman BI, Langefeld CD, et al. Apolipoprotein L1 gene variants associate with hypertension-attributed nephropathy and the rate of kidney function decline in African Americans. Kidney international 83 (2013): 114-120.
- Freedman BI, Kopp JB, Langefeld CD, et al. The apolipoprotein L1 (APOL1) gene and nondiabetic nephropathy in African Americans. Journal of the American Society of Nephrology: JASN 21 (2010): 1422-1426.
- Nqebelele NU, Dickens C, Dix-Peek T, et al. Low prevalence of apolipoprotein L1 gene variants in Black South Africans with hypertension-attributed chronic kidney disease. Clinical nephrology (2018).
- Wasser WG, Tzur S, Wolday D, et al. Population genetics of chronic kidney disease: the evolving story of APOL1. J Nephrol 25 (2012): 603-618.
- Genovese G, Friedman DJ, Ross MD, et al. Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science (New York, NY) 329 (2010): 841-845.
- Vanhollebeke B, Pays E. The function of apolipoproteins L. Cell Mol Life Sci 63 (2006): 1937-1944.
- Hoy WE, Hughson MD, Kopp JB, Mott SA, et al. APOL1 Risk Alleles Are Associated with Exaggerated Age-Related Changes in Glomerular Number and Volume in African-American Adults: An Autopsy Study. Journal of the American Society of Nephrology: JASN 26 (2015): 3179-3189.
- Foster MC, Coresh J, Fornage M, et al. APOL1 variants associate with increased risk of CKD among African Americans. Journal of the American Society of Nephrology: JASN 24 (2013): 1484-1491.
- Splinter K, Adams DR. Effect of Genetic Diagnosis on Patients with Previously Undiagnosed Disease 379 (2018): 2131-2139.
- Lv L, Wang J, Gao B, et al. Serum uromodulin and progression of kidney disease in patients with chronic kidney disease. J Transl Med 16 (2018): 316.
- Dahan K, Devuyst O, Smaers M, et al. A cluster of mutations in the UMOD gene causes familial juvenile hyperuricemic nephropathy with abnormal expression of uromodulin. Journal of the American Society of Nephrology : JASN 14 (2003): 2883-2893.
- Hart TC, Gorry MC, Hart PS, et al. Mutations of the UMOD gene are responsible for medullary cystic kidney disease 2 and familial juvenile hyperuricaemic nephropathy. J Med Genet 39 (2002): 882-892.
- Kottgen A, Hwang SJ, Larson MG, et al. Uromodulin levels associate with a common UMOD variant and risk for incident CKD. Journal of the American Society of Nephrology: JASN 21 (2010): 337-344.
- Kottgen A, Glazer NL, Dehghan A, et al. Multiple loci associated with indices of renal function and chronic kidney disease. Nat Genet 41 (2009): 712-717.
- Trudu M, Janas S, Lanzani C, et al. Common noncoding UMOD gene variants induce salt-sensitive hypertension and kidney damage by increasing uromodulin expression. Nat Med 19 (2013): 1655-1660.
- Parving HH, Smidt UM, Hommel E, et al. Effective antihypertensive treatment postpones renal insufficiency in diabetic nephropathy. American journal of kidney diseases : the official journal of the National Kidney Foundation 22 (1993): 188-195.
- Kasiske BL. Relationship between vascular disease and age-associated changes in the human kidney. Kidney international 31 (1987): 1153-1159.


 Impact Factor: * 3.1
Impact Factor: * 3.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
