Nisin, A Probiotic Bacteriocin, Combined with Limited Chemoradiation Therapy in Head and Neck Cancer
Wendy Fu1, Yvonne L Kapila2*
1Department of Preventive and Restorative Dental Sciences, University of California, San Francisco, USA
2Department of Orofacial Sciences, University of California, San Francisco, USA
*Corresponding Author: Yvonne L Kapila, Professor and Chair of Periodontology, Division of Periodontology, Department of Orofacial Sciences, University of California, San Francisco, 513 Parnassus Avenue, S616C, Box 0422, San Francisco, CA 94143, USA>
Received: 27 April 2021; Accepted: 14 June 2021; Published: 21 July 2021
Article Information
Citation: Wendy Fu, Yvonne L Kapila. Nisin, A Probiotic Bacteriocin, Combined with Limited Chemoradiation Therapy in Head and Neck Cancer. Archives of Clinical and Medical Case Reports 5 (2021): 531-536.
View / Download Pdf Share at FacebookAbstract
Objective: In this case report, we describe the use of nisin Z as an adjunctive therapy in a patient diagnosed with metastatic oropharyngeal cancer.
Case Report: A 57-year-old Caucasian male patient affected by metastatic oropharyngeal cancer, was prescribed neoadjuvant chemotherapy followed by weekly cisplatin chemotherapy with radiation therapy. During the initial phase of the chemoradiation therapy, the patient was hospitalized for swelling and edema in the throat and experienced other negative side effects, including nausea. The patient elected to stop recommended treatment and began taking nisin Z for 19 weeks. The patient has reported improved symptoms, including absence of pain, dysphagia, or dyspnea, and the tumor diminishing in size since taking nisin Z.
Results: Nisin Z may have contributed to improvement in the patient’s symptoms, including shrinkage of the tumor and extended survival.
Keywords
<p>Nisin; Probiotic bacteriocin; Neck cancer; Chemoradiation therapy</p>
Article Details
1. Introduction
Head and neck squamous cell carcinoma (HNSCC) is cancer of the head and neck and usually begins in the squamous cells that line the mucosal surfaces. Head and neck cancer is a devastating disease, often disfiguring and debilitating affected patients. It is the sixth most common cancer worldwide and comprises cancers of the oral cavity, larynx, hypopharynx, oropharynx, paranasal sinuses and nasal cavity, and salivary glands [1, 2]. In 2018, head and neck cancer accounted for approximately 706,000 new cases and 358,000 deaths worldwide [1]. In the US, head and neck cancer accounts for 3% of all cancers and approximately 65,000 Americans are diagnosed with head and neck cancer annually [1, 3, 4]. Alcohol and tobacco use are important risk factors for HNSCC, accounting for causing 75% of head and neck cancer [5]. Importantly, the 5-year survival rates for oral cancer are improving but remain <70%, depending on treatment, comorbidity, etc [6], underscoring the need for novel treatment approaches.
Nisin, an antimicrobial peptide produced by the probiotic Lactococcus lactis, may be such a treatment. Nisin was first used in the food industry as a natural biopreservative due to its antimicrobial properties [7, 8]. Nisin mediates antimicrobial effects by altering the integrity of cellular membranes and forming short-lived pores, resulting in changes in membrane potential [9]. In addition to its antibacterial properties, nisin has been examined for its pro-apoptotic properties against cancer cells [13-16]. Nisin and nisin Z, a naturally occurring variant of nisin, have been used to treat gastrointestinal infections [17], mastitis [18, 19], oral candidiasis [20, 21], and oral and head and neck squamous cell carcinoma [15, 16, 22, 23]. Although its effects on oral cancer have not been investigated in humans. This is the first reported case of nisin used to treat humans in a cancer setting.
2. Case Report
We report the case of a 57-year-old Caucasian male patient, affected by metastatic oropharyngeal cancer. Referred comorbidities were depression and history of seizure. The patient reported he smoked one pack of cigarettes per day for 45 years and quit smoking in 2014. The patient also reported he drank two six-packs or more of beer every day for 45 years and quit using alcohol in 2014. The patient reported his father passed away from HNSCC. In October 2017, the patient was diagnosed with basaloid squamous cell carcinoma at the base of the tongue with invasion to lymph nodes on the ipsilateral cervical area but with no distant disease (T3, N2a, M0) (Figure 1). The patient was scheduled for neoadjuvant chemotherapy (Taxotere, cisplatin, 5-FU) followed by weekly cisplatin chemotherapy with radiation therapy (6 MV linear accelerator with custom MLC blocking with an IMRT, to total dose of 7020 cGy, using 180 cGy fractions 5 days a week).
In early January 2018, the patient completed his first cycle of neoadjuvant chemotherapy. Two days later, he develo-ped swelling and edema in the throat and was admitted to the intensive care unit, where tracheotomy was performed. The patient was dismissed from the hospital two days later, and the cause of the acute swelling was not determined but was hypothesized as fast progression of the tumor. During post-operative evaluation, patient elected to have the tracheotomy tube removed although not recommended. In late January 2018, the patient completed his second round of neoadjuvant chemotherapy and was instructed to begin his radiation therapy with concomitant weekly chemotherapy (cisplatin), which the patient did not begin until late April 2018. By mid-May 2018, the patient had received 2880 cGy of a planned 7020 cGy for radiation therapy and 16 fractions of a planned 39 fractions of chemotherapy. Following this, the patient elected to stop recommended chemotherapy and radiation therapy due to side effects, including nausea.
In September 2018, after reading about publications regarding nisin anticancer effects for oral and head and neck cancer [15, 16], the patient on his own began taking high content nisin Z (Handary) in the form of powder (4.5g) mixed in water as a shake for 19 weeks. Following this, the patient continued taking nisin several times a week and stopped nisin around January 2021. The patient denies having pain, dysphagia, or dyspnea, and reports the tumor has diminished in size since he began taking nisin Z (Figure 2). The patient has since declined to see a doctor or specialist to monitor the disease progression, and he has returned to work as an operator in demolition services.
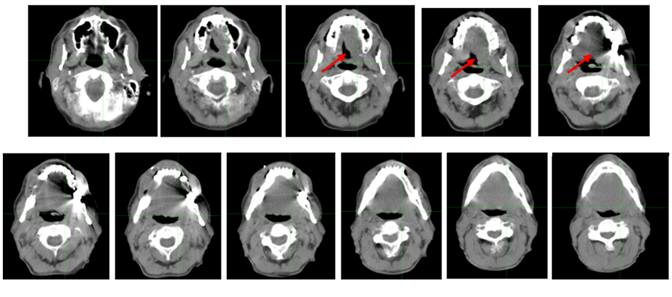
Figure 1: Images from patient's CT scan in November 2017, before chemoradiation therapy. Red arrows indicate location of tumor, as viewed by asymmetry of soft tissue.
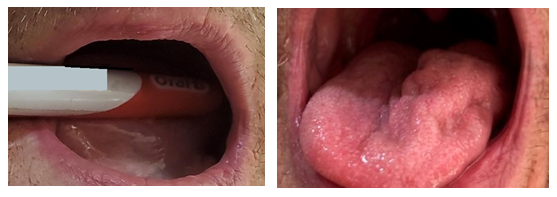
Figure 2: Intraoral photos taken by patient in April 2021, after 19 weeks of nisin Z treatment.
3. Discussion
We report here the case of a patient diagnosed with basaloid squamous cell carcinoma at base of the tongue who responded to nisin Z with reducing signs and symptoms of the disease. The patient completed a portion of the prescribed chemoradiation therapy prior to halting treatment and using nisin Z. Thus, it is difficult to separate the effects of chemoradiation therapy from anti-cancer effects of nisin. However, we interpret the improvement in the patient’s symptoms, including shrinkage of tumor and extended survival, as potential beneficial effects of nisin. Nisin is not toxic to animals and is safe for human consumption [24]. Nisin is commonly used in the food industry as a broad-spectrum bacteriocin against Gram-positive foodborne bacteria [24-26]. Recently, other properties of nisin, including its pro-apoptotic anticancer properties, have been studied. Cancer cells are resistant to apoptosis.
However, due to differences in cell membrane composition of HNSCC cells compared to primary keratinocytes [27, 28], nisin may preferentially affect cancer cell membranes and induce pore formation that leads to a net influx of ions, including calcium, and eventual cell death. Calcium plays a key role in inducing cell death via formation of the apoptosome and activation of cell surface death receptors and caspases. There has been no clinical trial reporting the use of nisin to treat HNSCC. The only reported clinical use of nisin in humans was to treat mastitis during lactation [29]. However, previous reports include in vitro and animal studies that demonstrated positive anti-cancer effects of nisin on HNSCC [15, 16, 22, 23]. To our knowledge, this is the first case report in the literature illustrating the potential anti-cancer effects of nisin in humans.
4. Conclusion
We speculate that nisin Z contributed to the reduction of the tumor in this patient via its pro-apoptotic properties on cancer cells. Therefore, the therapeutic effects of nisin in treating HNSCC in patients should be further investigated, as it could be a safe adjunctive therapy to traditional chemoradiation therapy, with limited side effects.
References
- Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68 (2018): 394-424.
- Jethwa AR, Khariwala SS. Tobacco-related carcinogenesis in head and neck cancer. Cancer Metastasis Rev 36 (2017): 411-423.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin 70 (2020): 7-30.
- Fitzmaurice C, Allen C, Barber RM, et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol 3 (2017): 524-548.
- Blot WJ, McLaughlin JK, Winn DM, et al. Smoking and drinking in relation to oral and pharyngeal cancer. Cancer Research 48 (1988): 3282-3287.
- Surveillance Research Program, National Cancer Institute SEER*Stat software (seer.cancer.gov/seerstat) version <1975-2017>.
- Lubelski J, Rink R, Khusainov R, et al. Biosynthesis, immunity, regulation, mode of action and engineering of the model lantibiotic nisin. Cell Mol Life Sci 65 (2008): 455-476.
- De Arauz LJ, Jozala AF, Mazzola PG, et al. Nisin biotechnological production and application: a review. Trends Food Sci Technol 20 (2009): 146-154.
- Moll GN, Clark J, Chan WC, et al. Role of transmembrane pH gradient and membrane binding in nisin pore formation. J Bacteriol 179 (1997): 135-140.
- Nonzee NJ, Dandade NA, Patel U, et al. Evaluating the supportive care costs of severe radiochemotherapy-induced mucositis and pharyngitis: results from a Northwestern University Costs of Cancer Program pilot study with head and neck and nonsmall cell lung cancer patients who received care at a county hospital, a Veterans Administration hospital, or a comprehensive cancer care center. Cancer 113 (2008): 1446-1452.
- Bsoul SA, Huber MA, Terezhalmy GT. Squamous cell carcinoma of the oral tissues: a comprehensive review for oral healthcare providers. J Contemp Dent Pract 6 (2005): 1-16.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin 62 (2012): 10-29.
- Farkas-Himsley H, Zhang YS, Yuan M, et al. Partially purified bacteriocin kills malignant cells by apoptosis: programmed cell death. Cell Mol Biol (Noisy-le-grand) 38 (1992): 643-651.
- Shin JM, Ateia I, Paulus JR, et al. Antimicrobial nisin acts against saliva derived multi-species biofilms without cytotoxicity to human oral cells. Front Microbiol 6 (2015): 617.
- Joo NE, Ritchie K, Kamarajan P, et al. Nisin, an apoptogenic bacteriocin and food preservative, attenuates HNSCC tumorigenesis via CHAC1. Cancer Med 1 (2012): 295-305.
- Kamarajan P, Hayami T, Matte B, et al. Nisin ZP, a Bacteriocin and Food Preservative, Inhibits Head and Neck Cancer Tumorigenesis and Prolongs Survival. PLoS One 10 (2015): e0131008.
- Blay GL, Lacroix C, Zihler A, et al. In vitro inhibition activity of nisin A, nisin Z, pediocin PA-1 and antibiotics against common intestinal bacteria. Lett Appl Microbiol 45 (2007): 252-257.
- Cao LT, Wu JQ, Xie F, et al. Efficacy of nisin in treatment of clinical mastitis in lactating dairy cows. J Dairy Sci 90 (2007): 3980-3985.
- Wu J, Hu S, Cao L. Therapeutic effect of nisin Z on subclinical mastitis in lactating cows. Antimicrob Agents Chemother 51 (2007): 3131-3135.
- Le Lay C, Akerey B, Fliss I, et al. Nisin Z inhibits the growth of Candida albicans and its transition from blastospore to hyphal form. J Appl Microbiol 105 (2008): 1630-1639.
- Akerey B, Le Lay C, Fliss I, et al. In vitro efficacy of nisin Z against Candida albicans adhesion and transition following contact with normal human gingival cells. J Appl Microbiol 107 (2009): 1298-1307.
- Kamarajan P, Ateia I, Shin JM, et al. Periodontal pathogens promote cancer aggressivity via TLR/MyD88 triggered activation of Integrin/FAK signaling that is therapeutically reversible by a probiotic bacteriocin. PLoS Pathog 16 (2020): e1008881.
- Shin JM, Gwak JW, Kamarajan P, et al. Biomedical applications of nisin. J Appl Microbiol 120 (2016): 1449-1465.
- Cleveland J, Montville TJ, Nes IF, et al. Bacteriocins: safe, natural antimicrobials for food preservation. Int J Food Microbiol 71 (2001): 1-20.
- Delves-Broughton J, Blackburn P, Evans RJ, et al. Applications of the bacteriocin, nisin. Antonie Van Leeuwenhoek 69 (1996): 193-202.
- Severina E, Severin A, Tomasz A. Antibacterial efficacy of nisin against multidrug-resistant Gram-positive pathogens. J Antimicrob Chemother 41 (1998): 341-347.
- Tripathi P, Kamarajan P, Somashekar BS, et al. Delineating metabolic signatures of head and neck squamous cell carcinoma: phospholipase A2, a potential therapeutic target. Int J Biochem Cell Biol 44 (2012): 1852-1861.
- Somashekar BS, Kamarajan P, Danciu T, et al. Magic angle spinning NMR-based metabolic profiling of head and neck squamous cell carcinoma tissues. J Proteome Res 10 (2011): 5232-5241.
- Fernandez L, Delgado S, Herrero H, et al. The bacteriocin nisin, an effective agent for the treatment of staphylococcal mastitis during lactation. J Hum Lact 24 (2008): 311-316.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
