A Case of Adult Onset Neuronal Intranuclear Inclusion Disease
Mingyuan Xu, Jianzhong Wang, Dazhen Cha, Pengxin Lin, Zhixiong Zheng*
Mindong Hospital Affiliated to Fujian Medical University, Fuan, Fujian, China
*Corresponding Author: Dr. Zhixiong Zheng, Mindong Hospital Affiliated to Fujian Medical University, Fuan, Fujian, China
Received: 19 August 2019; Accepted: 12 September 2019; Published: 26 November 2019
Article Information
Citation: Zhixiong Zheng, Mingyuan Xu, Jianzhong Wang, Dazhen Cha, Pengxin Lin. A Case of Adult Onset Neuronal Intranuclear Inclusion Disease. Archives of Clinical and Medical Case Reports 3 (2019): 605-610.
View / Download Pdf Share at FacebookAbstract
In this case of adult onset neuronal intranuclear inclusion disease (NIID), we will explore the clinical and pathological features of NIID as well as its medical imaging and histological features. The patient presented with tremor, cognitive impairment, and paroxysmal disturbance of consciousness. Diffusion-weighted imaging of cranial magnetic resonance showed high signal intensity of ribbon-like distribution at corticomedullary junction. Additionally, fibloblasts, vascular endothelial cells, and intranuclear inclusions were found on histological specimens of skin biopsy. This case suggests adult onset NIID is a disease of chronic neurological degeneration with high heterogeneity, which can be diagnosed by characteristic MRI DW1 ribbon-like high signal and eosinophilic intranuclear inclusions under electron microscopy.
Keywords
Neuronal Intranuclear Inclusion Disease, Dementia; Ribbon-like high signal, Intranuclear inclusions
Neuronal Intranuclear Inclusion Disease articles, Dementia articles, Ribbon-like high signal articles, Intranuclear inclusions articles
Neuronal Intranuclear Inclusion Disease articles Neuronal Intranuclear Inclusion Disease Research articles Neuronal Intranuclear Inclusion Disease review articles Neuronal Intranuclear Inclusion Disease PubMed articles Neuronal Intranuclear Inclusion Disease PubMed Central articles Neuronal Intranuclear Inclusion Disease 2023 articles Neuronal Intranuclear Inclusion Disease 2024 articles Neuronal Intranuclear Inclusion Disease Scopus articles Neuronal Intranuclear Inclusion Disease impact factor journals Neuronal Intranuclear Inclusion Disease Scopus journals Neuronal Intranuclear Inclusion Disease PubMed journals Neuronal Intranuclear Inclusion Disease medical journals Neuronal Intranuclear Inclusion Disease free journals Neuronal Intranuclear Inclusion Disease best journals Neuronal Intranuclear Inclusion Disease top journals Neuronal Intranuclear Inclusion Disease free medical journals Neuronal Intranuclear Inclusion Disease famous journals Neuronal Intranuclear Inclusion Disease Google Scholar indexed journals Disease articles Disease Research articles Disease review articles Disease PubMed articles Disease PubMed Central articles Disease 2023 articles Disease 2024 articles Disease Scopus articles Disease impact factor journals Disease Scopus journals Disease PubMed journals Disease medical journals Disease free journals Disease best journals Disease top journals Disease free medical journals Disease famous journals Disease Google Scholar indexed journals Dementia articles Dementia Research articles Dementia review articles Dementia PubMed articles Dementia PubMed Central articles Dementia 2023 articles Dementia 2024 articles Dementia Scopus articles Dementia impact factor journals Dementia Scopus journals Dementia PubMed journals Dementia medical journals Dementia free journals Dementia best journals Dementia top journals Dementia free medical journals Dementia famous journals Dementia Google Scholar indexed journals Ribbon-like high signal articles Ribbon-like high signal Research articles Ribbon-like high signal review articles Ribbon-like high signal PubMed articles Ribbon-like high signal PubMed Central articles Ribbon-like high signal 2023 articles Ribbon-like high signal 2024 articles Ribbon-like high signal Scopus articles Ribbon-like high signal impact factor journals Ribbon-like high signal Scopus journals Ribbon-like high signal PubMed journals Ribbon-like high signal medical journals Ribbon-like high signal free journals Ribbon-like high signal best journals Ribbon-like high signal top journals Ribbon-like high signal free medical journals Ribbon-like high signal famous journals Ribbon-like high signal Google Scholar indexed journals Intranuclear inclusions articles Intranuclear inclusions Research articles Intranuclear inclusions review articles Intranuclear inclusions PubMed articles Intranuclear inclusions PubMed Central articles Intranuclear inclusions 2023 articles Intranuclear inclusions 2024 articles Intranuclear inclusions Scopus articles Intranuclear inclusions impact factor journals Intranuclear inclusions Scopus journals Intranuclear inclusions PubMed journals Intranuclear inclusions medical journals Intranuclear inclusions free journals Intranuclear inclusions best journals Intranuclear inclusions top journals Intranuclear inclusions free medical journals Intranuclear inclusions famous journals Intranuclear inclusions Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals microscopy articles microscopy Research articles microscopy review articles microscopy PubMed articles microscopy PubMed Central articles microscopy 2023 articles microscopy 2024 articles microscopy Scopus articles microscopy impact factor journals microscopy Scopus journals microscopy PubMed journals microscopy medical journals microscopy free journals microscopy best journals microscopy top journals microscopy free medical journals microscopy famous journals microscopy Google Scholar indexed journals antagonist articles antagonist Research articles antagonist review articles antagonist PubMed articles antagonist PubMed Central articles antagonist 2023 articles antagonist 2024 articles antagonist Scopus articles antagonist impact factor journals antagonist Scopus journals antagonist PubMed journals antagonist medical journals antagonist free journals antagonist best journals antagonist top journals antagonist free medical journals antagonist famous journals antagonist Google Scholar indexed journals infusion articles infusion Research articles infusion review articles infusion PubMed articles infusion PubMed Central articles infusion 2023 articles infusion 2024 articles infusion Scopus articles infusion impact factor journals infusion Scopus journals infusion PubMed journals infusion medical journals infusion free journals infusion best journals infusion top journals infusion free medical journals infusion famous journals infusion Google Scholar indexed journals patient articles patient Research articles patient review articles patient PubMed articles patient PubMed Central articles patient 2023 articles patient 2024 articles patient Scopus articles patient impact factor journals patient Scopus journals patient PubMed journals patient medical journals patient free journals patient best journals patient top journals patient free medical journals patient famous journals patient Google Scholar indexed journals
Article Details
1. Introduction
Neuronal intranuclear inclusion disease (NIID) is a rare neurodegenerative disorder characterized pathologically by the presence of eosinophilic intranuclear inclusions in neuronal and glial cells. It has been considered to be a heterogeneous disease entity because the clinical pictures of previously described cases were highly variable [1]. Patients with NIID present with a variety of symptoms including pyramidal and extra-pyramidal signs, ataxia, dementia, epileptic seizure, peripheral neuropathy and autonomic dysfunction. Head magnetic resonance imaging (MRI) and skin biopsy have important value in diagnosing NIID. The case is reported as neuronal intranuclear inclusion disease (NIID) with main clinical features including tremor, cognitive impairment and paroxysmal disturbance of consciousness. We analyzed the clinical and pathological features as well as medical imaging in order to raise awareness of NIID.
2. Case Report
2.1 Medical History
Mrs Luo is a 76 yo female with a history of tremor and paroxysmal disturbance of consciousness who was admitted to hospital with two days of confusion and difficulty communicating on Feburary 20, 2019. The tremor began in 2006. Initially, it was in the upper extremities, and the symptoms became aggravated when she carried objects by hands. Occasionally, it happened in the lower extremities, and it fluctuated. She also had impaired memory, mainly recent memory impairement, including being forgetful, without a sense of place. In May 2011, she felt disturbance of consciousness and rambling in her speech with feverish sensation but without paralysis and convulsion. She was afebrile, with normal blood sugar and blood pressure. The above symptoms continued to disappear for about 1 day. Since October 2011 she had recurrent disturbance of consciousness which manifested the above-mentioned symptoms, rambling in her speech, as well as sensory hallucination and being unable to communicate with others after feeling feverish sensation. The symptoms last 4 days, then improved spontaneously, but her memory degraded. The symptoms would require up to five times each year, and she had been admitted to the local hospital for more than 10 times without a proper diagnosis.
On Feburary 20, 2019, she was reported feverish sensation, confusion of consciousness, difficulty articulating speech and communicating as well as urinary incontinence, no limb weakness, no seizure and movement disorder.
Past Medical History: denied hypertension, DM, no tuberculosis, syphilis, denies transfusion and lead poison history.
2.2 Allergy
No known allergy.
2.3 Family History
Her brother and niece had upper limb motor tremor.
2.4 Admission examination
T 36.5°C, HR 80 beats/min, RR 18 breathes/min, BP134/88.
Drowsiness, reduced consciousness, mixed aphasia, patient could not cooperate with neurological examination, pupils are equal and reactive to light (diameter approx. 1.5 mm), extraocular eye movements intact, bilateral nasolabial folds are symmetrical, and the tongue is not cooperative. Neck soft; Both lungs are clear; Regular heart rate and rhythm, no murmurs. Abdominal soft, no tenderness, liver and spleen not touched. Muscle strength cannot cooperate, but painful stimulation can be lifted off the bed surface, limb muscle tension is normal, quadriplegia tendon reflex, bilateral palmar reflexes positive, bilateral Babinski sign negative, mutual aid exercise, deep sensory examination cannot cooperate.
2.5 Laboratory data
blood routine, urine routine, liver and kidney function, blood sugar, glycosylated hemoglobin, and blood homocysteine ??are normal. Thyroid function and thyroid peroxidase antibodies, tumor markers, immune complexes, and anti-neutrophil cytoplasmic antibodies (ANCA) are all in the normal range. Blood RPR, TPPA, HIV antibody negative. Cerebrospinal fluid routine and biochemical examination results are normal. Cerebrospinal fluid-based cytology: no significant increase in cell mass, see a small number of neutrophils, lymphocytes; no atypical cells. Figure 1A and 1B shows cerebral magnetic resonance DWI showed bilateral symmetry ribbon-like high signal distribution at the junction of the frontal, apical, temporal and occipital lobe. Figure 2 shows EEG of the anterior head of both sides saw the 3-4 Hz slow wave in the burst (Figure 2).
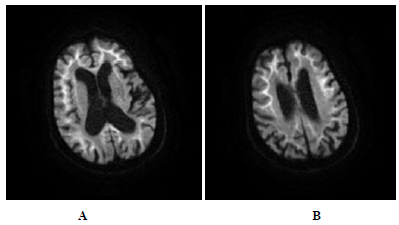
Figure 1A and B: Cerebral magnetic resonance DWI showed bilateral symmetry ribbon-like high signal distribution at the junction of the frontal, apical, temporal and occipital lobe (Figure 1A and 1B).
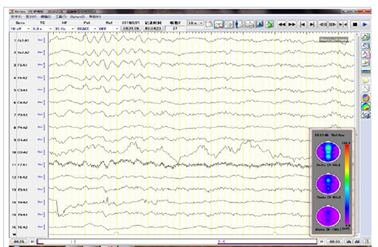
Figure 2: EEG of the anterior head of both sides saw the 3-4 Hz slow wave in the burst.
Skin biopsy light microscopy: "right calf skin biopsy": skin tissue examination: hyperkeratosis of the epidermis, thickening of the local acanthosis, papillary projection, collagenization of the superficial dermis (Figure 3); electron microscopy: a few fibroblasts, vascular endothelial cells, circular inclusion bodies in the nucleus of cells, and the size of each was unequal, with a diameter of 0.2-0.6 μm and a circular non-enveloped filament structure (Figure 4). Uroelectron microscopy: Partially exfoliated cells of the urine were ruptured, and no typical inclusion body-like structure was found in the residual nucleus and leukocyte nucleus, individual small nerve bundles could be seen, and the density of myelinated nerves is reduced (Figure 5A, 5B).
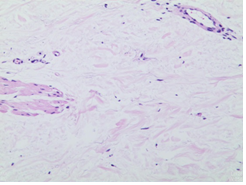
Figure 3: Skin biopsy light microscopy: "right calf skin biopsy": skin tissue examination: hyperkeratosis of the epidermis, thickening of the local acanthosis, papillary projection, collagenization of the superficial dermis.
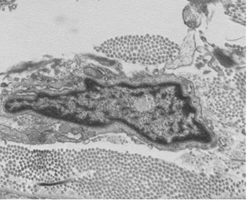
Figure 4: Electron microscopy of cells shows a few fibroblasts, vascular endothelial cells, circular inclusion bodies in the nucleus.
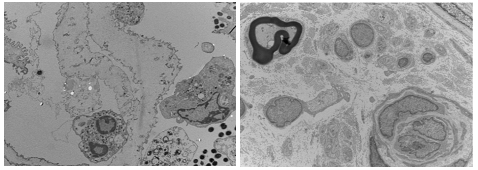
Figure 5: Uroelectron microscopy: Partially exfoliated cells of the urine were ruptured, and no typical inclusion body-like structure was found in the residual nucleus and leukocyte nucleus, individual small nerve bundles could be seen, and the density of myelinated nerves is reduced.
3. Treatment and Evolution of the Disease
symptomatic supportive treatment was given after admission. On the third day, the patient's consciousness was cleared and she began to understand other people's speech. On the 6th day of admission, the patient was able to take care of herself but still had memory loss. Neurological examination: clear speech, bilateral pupils, and other large circles, D ≈ 2.0 mm, light response, eye movements, limb muscle strength, muscle tension normal, Romberg's sign positive. MMSE score: 14 points.
4. Discussion
NIID is considered to be a heterogeneous disease with highly variable clinical manifestations, including neuropathy, cerebellar ataxia, and dementia [2]. In 2003, Takahashi Fujigasaki [1] classified NIID into infant type, adolescent type and adult type according to the age of onset, and was divided into sporadic and family type according to hereditary methods. In adult-onset NIID, most cases are diagnosed at 60 to 70 years of age [3].
Our patient showed tremble, cognitive impairment, paroxysmal disturbance of consciousness, and autonomic nerve damage, similar to the tremor seen in dementia with Lewy bodies (DLB). The patients with DLB could present fluctuating cognition, visual hallucinations, REM sleep behavior disorder, signs of parkinsonism, and autonomic dysfunction [4]. However, REM sleep behavior disorder is not seen in this case. Parkinson’s disease dementia (PDD) starts as a movement disorder, but progresses to dementia and changes in mood and behavior [5]. However, our patient starts as tremble, but no other movement disorder. Dementia can also caused by Alzheimer’s disease, vascular dementia, normal pressure hydrocephalus [6]. However, symptoms including tremble, paroxysmal disturbance of consciousness, and autonomic nerve damage are rarely seen in dementia caused by the above-mentioned diseases. Paroxysmal disturbance of consciousness may mimic delilium. But with careful assessment and history, we can exclude delilum caused by uninary tract infection, hypoglycemic encephalopathy, Lupus erythematosus encephalopathy, side effect of a medication, withdrawal from drugs [7].
Sone et al. [8] reported that high signal intensity in the corticomedullary junction in DWI was a highly characteristic finding in NIID, and skin biopsy was useful for diagnosis. We also made a diagnosis of NIID based on the findings of MRI and skin biopsy. These previous reports and the present case suggest that NIID patients can develop dementia that is similar to that seen in DLB. Therefore, NIID should be considered in the differential diagnosis of dementia.
References
- Takahashi-Fujigasaki J. Neuronal intranuclear hyaline inclusion disease. Neuropathology 23 (2003): 351-359.
- Sone J, Kitagawa N, Sugawara E. Neuronal intranuclear inclusion disease cases with leukoencephalopathy diagnosed via skin biopsy. J. Neurol. Neurosurg. Psychiatry 85 (2014): 354-356.
- Takahashi-Fujigasaki J, Nakano Y, Uchino A. Adult-onset neuronal intranuclear hyaline inclusion disease is not rare in older adults. Geriatr Gerontol Int 16 (2016): 51-56.
- McKeith IG, Boeve BF, Dickson DW, et al. Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium". Neurology (Review). 89 (2017): 88-100.
- Sveinbjornsdottir S. The clinical symptoms of Parkinson's disease". Journal of Neurochemistry. 139 (2016): 318-324.
- Burns A, Iliffe S. Dementia. BMJ 338 (2009): b75.
- Gleason OC. Delirium. American Family Physician 67 (2003): 1027-1034.
- Sone J, Kitagawa N, Sugawara E, et al. Neuronal intranuclear inclusion disease cases with leukoencephalopathy diagnosed via skin biopsy. J Neurol Neurosurg Psychiatry 85 (2014): 354-356.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
