LCH with Multisystemic Involvement-A Case Report
Shreya Sehgal MD, Muniraju Maralakunte MD, Lokesh Singh MD, Uma Debi MD*
Department of Radiodiagnosis and Imaging, PGIMER, Chandigarh, India
*Corresponding Author: Uma Debi MD, Department of Radiodiagnosis and Imaging, PGIMER, Chandigarh, India
Received: 26 October 2020; Accepted: 07 December 2020; Published: 04 January 2021
Article Information
Citation: Shreya Sehgal, Muniraju Maralakunte, Lokesh Singh, Uma Debi. LCH with Multisystemic Involvement-A Case Report. Archives of Clinical and Medical Case Reports 5 (2021): 43-47.
View / Download Pdf Share at FacebookAbstract
Langerhans cell histiocytosis (LCH) constitutes a rare group of disorder derived from the macrophages and dendritic cells. The cells in LCH show close similarity to the langerhans cells found in the skin and mucosa and stain positive for CD1a, S100, and CD207 antigens. These cells display the same intracytoplasmic organelles on electron microscopy as seen in Langerhans cells i.e. Birbeck granules. LCH is ten times more prevalent in the paediatric age group as compared to adult population. Females are affected more commonly as compared to the males. LCH can have single- or multi-system types of involvement. Unifocal or multifocal pattern is seen in singlesystem type of disease. The multisystem pattern is often seen in the paediatric population while unisystem pattern is seen in adults. We present case report of a 35 yrs old male patient with multisystemic involvement.
Keywords
LCH; Bones; Lungs cysts; Liver; Skull
LCH articles; Bones articles; Lungs cysts articles; Liver articles; Skull articles
LCH articles LCH Research articles LCH review articles LCH PubMed articles LCH PubMed Central articles LCH 2023 articles LCH 2024 articles LCH Scopus articles LCH impact factor journals LCH Scopus journals LCH PubMed journals LCH medical journals LCH free journals LCH best journals LCH top journals LCH free medical journals LCH famous journals LCH Google Scholar indexed journals Bones articles Bones Research articles Bones review articles Bones PubMed articles Bones PubMed Central articles Bones 2023 articles Bones 2024 articles Bones Scopus articles Bones impact factor journals Bones Scopus journals Bones PubMed journals Bones medical journals Bones free journals Bones best journals Bones top journals Bones free medical journals Bones famous journals Bones Google Scholar indexed journals Lungs cysts articles Lungs cysts Research articles Lungs cysts review articles Lungs cysts PubMed articles Lungs cysts PubMed Central articles Lungs cysts 2023 articles Lungs cysts 2024 articles Lungs cysts Scopus articles Lungs cysts impact factor journals Lungs cysts Scopus journals Lungs cysts PubMed journals Lungs cysts medical journals Lungs cysts free journals Lungs cysts best journals Lungs cysts top journals Lungs cysts free medical journals Lungs cysts famous journals Lungs cysts Google Scholar indexed journals Liver articles Liver Research articles Liver review articles Liver PubMed articles Liver PubMed Central articles Liver 2023 articles Liver 2024 articles Liver Scopus articles Liver impact factor journals Liver Scopus journals Liver PubMed journals Liver medical journals Liver free journals Liver best journals Liver top journals Liver free medical journals Liver famous journals Liver Google Scholar indexed journals Skull articles Skull Research articles Skull review articles Skull PubMed articles Skull PubMed Central articles Skull 2023 articles Skull 2024 articles Skull Scopus articles Skull impact factor journals Skull Scopus journals Skull PubMed journals Skull medical journals Skull free journals Skull best journals Skull top journals Skull free medical journals Skull famous journals Skull Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals Langerhans cell histiocytosis articles Langerhans cell histiocytosis Research articles Langerhans cell histiocytosis review articles Langerhans cell histiocytosis PubMed articles Langerhans cell histiocytosis PubMed Central articles Langerhans cell histiocytosis 2023 articles Langerhans cell histiocytosis 2024 articles Langerhans cell histiocytosis Scopus articles Langerhans cell histiocytosis impact factor journals Langerhans cell histiocytosis Scopus journals Langerhans cell histiocytosis PubMed journals Langerhans cell histiocytosis medical journals Langerhans cell histiocytosis free journals Langerhans cell histiocytosis best journals Langerhans cell histiocytosis top journals Langerhans cell histiocytosis free medical journals Langerhans cell histiocytosis famous journals Langerhans cell histiocytosis Google Scholar indexed journals surgery articles surgery Research articles surgery review articles surgery PubMed articles surgery PubMed Central articles surgery 2023 articles surgery 2024 articles surgery Scopus articles surgery impact factor journals surgery Scopus journals surgery PubMed journals surgery medical journals surgery free journals surgery best journals surgery top journals surgery free medical journals surgery famous journals surgery Google Scholar indexed journals SARS-COV-2 articles SARS-COV-2 Research articles SARS-COV-2 review articles SARS-COV-2 PubMed articles SARS-COV-2 PubMed Central articles SARS-COV-2 2023 articles SARS-COV-2 2024 articles SARS-COV-2 Scopus articles SARS-COV-2 impact factor journals SARS-COV-2 Scopus journals SARS-COV-2 PubMed journals SARS-COV-2 medical journals SARS-COV-2 free journals SARS-COV-2 best journals SARS-COV-2 top journals SARS-COV-2 free medical journals SARS-COV-2 famous journals SARS-COV-2 Google Scholar indexed journals tomography articles tomography Research articles tomography review articles tomography PubMed articles tomography PubMed Central articles tomography 2023 articles tomography 2024 articles tomography Scopus articles tomography impact factor journals tomography Scopus journals tomography PubMed journals tomography medical journals tomography free journals tomography best journals tomography top journals tomography free medical journals tomography famous journals tomography Google Scholar indexed journals
Article Details
1. Case Report
A 35yr old male patient presented to our tertiary care hospital with complaints of pain abdomen, jaundice and a large swelling over the head. Plain radiographs of the head, chest and pelvis were done. Multiple punched out lytic calvarial lesions having bevelled margins were seen on skull radiographs. A large craniectomy defect was also noted on the left side of skull on the plain x-ray suggesting post-operative status (Figure 1 A-B). Plain radiograph of the chest showed diffuse reticular opacities in bilateral lungs with sparing of the bilateral costophrenic angles and well-defined lytic lesion with thin sclerotic rim was seen in left iliac bone, another small lytic lesion was seen in inferior part of right iliac bone, close to the right SI joint in the plain X-ray of the hip (Figure 1 C-D) . Further, contrast enhanced CT scan of the head, chest and abdomen was done to evaluate complete extent of the disease. Contrast enhanced CT scan of the head revealed lytic lesions in frontal and occipital bones on both sides with associated soft tissue component causing destruction of both inner and outer tables of skull. Scans through chest showed multiple variable sized bizarre shaped cysts in bilateral lung parenchyma with sparing of bilateral costophrenic angles (Figure 2 A-B). Scans through abdomen showed bilobar IHBRD with hypodense thickening seen at the level of primary biliary confluence (Figure 2 C-D). Lytic lesions were seen involving lower lumbar vertebral bodies and in the pelvis (Figure 2 E-F).
The patient underwent ultrasound-guided FNAC from this swelling, which revealed numerous mononuclear and many scattered bi- and multinucleated histiocytes having vesicular chromatin, grooved nuclei with coffee bean appearance and moderate to abundant amount of vacuolated cytoplasm. A provisional diagnosis of Langerhans cell histiocytosis was given. Subsequently, this calvarial lesion was removed surgically. Histopathological analysis of this specimen showed histiocytes, which stained positively for CD1a antigen. Following this, patient was started on chemotherapeutic agent Cladribine. He had no other significant past history or family history. Presently, the laboratory parameters of the patient were as follows: Total -1.85 mg/dl, conjugated -1.74 mg/dl bilirubin and serum ALP -1142U/l levels were elevated. Serum IgG levels were within normal range. Haemoglobin -10.6 gm/dl, total protein- 6.28 g/dL; albumin- 2.74 g/dL were mild reduced. Other parameters were within normal range: RBC count-3.6 × 1012/L, platelet count-318,000/mm3, AST- 40 IU/mL, ALT-37 IU/mL.
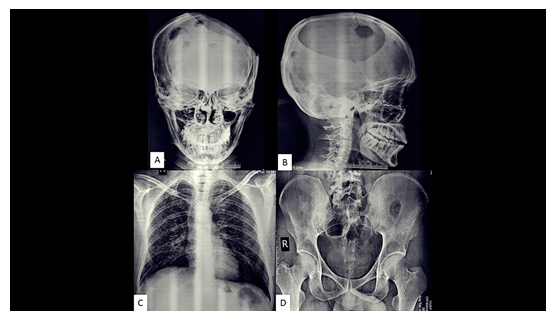
Figure 1: (A-D) AP and lateral plain skull radiographs showed few well circumscribed lesions with bevelled margins (A-B). A large craniectomy defect noted on the left side of head (post-op case). X-ray of the chest postero-anterior view showed reticular opacities in bilateral lungs (C); (D) X-ray of the pelvis with both hips anteroposterior view showed well-circumscribed lytic lesions in bilateral iliac bones.
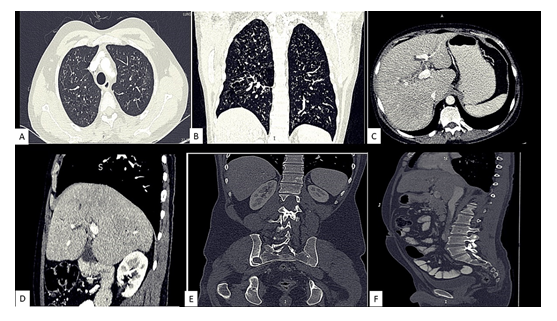
Figure 2: (A-F) Axial and Coronal reformatted (A-B) lung window images revealed innumerable bizarre shaped cysts equally distributed in bilateral lung parenchyma with sparing of bilateral costophrenic angles. Axial and sagittal reformatted (C-D) soft tissue window images showed ill-defined hypodensity along the biliary tracts at the level of primary biliary confluence with mild bilobar IHBRD. Coronal and sagittal reformatted bone window images show well circumscribed lytic lesions without sclerotic margins in the right side pelvis and in the L3 vertebral body respectively (E-F).
2. Discussion
In LCH, there is non-malignant proliferation of the monoclonal dendritic cells in one or multiple organs systems in the body. The annual incidence of the disease in paediatric age group is 2.6-5.4 cases per million [1-6]. However, the exact incidence in adult age group is not known due to rarity of this disease in adults [7, 8]. Both uni-system and multi-system patterns can be seen in adults. In our case, a multisystem pattern of involvement was observed with involvement of bone, lung and liver seen. Flat bones are involved more frequently than the long bones [9]. Calvarial involvement being the commonest. Typical finding is of a punched out lytic lesion with a bevelled edge due to unequal destruction of out and inner tables of skull. These calvarial lesions due not show any surrounding periosteal reaction [9]. However, bony lesions of LCH at other sites would show periosteal reaction. Early vertebral disease is seen as lytic lesions in the body of vertebra which later progresses to flattening of the vertebral body (vertebra plana). Intervertebral disc space is usually preserved. Long bones like femur, humerus and tibia are more typically involved in children. In the adult age group, plasmacytomas, brown tumours and metastasis form the close differentials.
Posterior pituitary and the pituitary stalk are commonest sites of involvement in the CNS, causing diabetes insipidus. Loss of posterior pituitary bright spot is seen on T1 weighted images. Thickening and enhancement of stalk is seen on post-contrast images [2]. Abnormal areas with white matter signal alteration can be seen in the cerebellum and in the basal ganglia appearing hyper-intense on the fluid sensitive sequences [10]. Lung involvement has a strong association with smoking and is commonly seen in adult patients with this disease [11]. Centrilobular nodules are seen in early stage of the disease. In the early stage, metastasis, miliary tuberculosis, sarcoidosis and silicosis form close differentials of this disease [12]. Late stage is characterised by cysts (usually <1 cm in size), which may coalesce to form large bullae. These bullae can rupture causing spontaneous pneumothorax. The upper and middle lobes are typically involved. Sparing of bilateral costophrenic angles is characteristic. Enlarged mediastinal or hilar lymph nodes or pleural effusion is not commonly seen [13]. Lymphangioleiomyomatosis (LAM) and lymphocytic interstitial pneumonia (LIP) are the main differentials [12]. The lung cysts are of similar size and morphology in LAM and background lung parenchyma is normal unlike LCH [14]. Hepatic involvement in the adult population is rare and is associated with poor prognosis [5]. Hepatomegaly and liver nodules characterize early liver disease. Late liver involvement is characterised by the fibrotic stage, which is mostly limited to the bile ducts and leads to progressive sclerosing cholangitis [15].
3. Conclusion
Our case represents adult multisystem variety of LCH, which is usually associated with poor prognosis. LCH should be kept in mind as a close differential if such a multisystem pattern of involvement is seen. Good results have been achieved with the use of chemotherapeutic agent Cladribine, which was initiated in our case.
Conflicts of Interests
None.
Acknowledgements
None.
References
- Jaffe R, Weiss LM, Facchetti F. Tumours derived from Langerhans cells. In Eds.: Swerdlow SH, Campo E, Harris NL, et al. World Health Organization classification of tumours of haematopoietic and lymphoid tissues. Lyon, France: IARC (2008): 358.
- Badalian-Very G, Vergilio JA, Fleming M, et al. Pathogenesis of Langerhans cell histiocytosis. Annu Rev Pathol 8 (2013): 1-20.
- Jaffe R. The histiocytoses. Clin Lab Med 19 (1999): 135-155.
- Arico M, Girschikofsky M, En´ Ereau TG, et al. Langerhans cell histiocytosis in adults. Report from the International Registry of the Histiocyte Society. European Journal of Cancer 39 (2003): 2341-2348.
- The French Langerhans Cell Histiocytosis Study Group. A multicentre retrospective survey of Langerhans’ cell histiocytosis: 348 cases observed between 1983 and 1993. Archives of Disease in Childhood 75 (1996): 17-24.
- Abla O, Egeler RM, and Weitzman S. Langerhans cell histiocytosis: current concepts and treatments. Cancer Treatment Reviews 36 (2010): 354-359.
- Broadbent V, Egeler R, Nesbit MJr. Langerhans cell histiocytosis–clinical and epidemiological aspects. Br J Cancer Suppl 23 (1994): 11-16.
- Bhatia S, Nesbit ME, Egeler M, et al. Epidemiologic study of Langerhans cell histiocytosis in children. J Pediatr 130 (1997): 774-784.
- Stull MA, Kransdorf MJ, Devaney KO. Langerhans cell histiocytosis of bone. RadioGraphics 12 (1992): 801-823.
- D’Ambrosio N, Soohoo S, Warshall C, et al. Craniofacial and intracranial manifestations of Langerhans cell histiocytosis: report of findings in 100 patients. AJR Am J Roentgenol 191 (2008): 589-597.
- Vassallo R, Ryu JH, Schroeder DR, et al. Clinical outcomes of pulmonary Langerhans’-cell histiocytosis in adults. N Engl J Med 346 (2002): 484-490.
- De Mello RA, Tanos JW, Mello MB, et al. Multisystemic Langerhans cell histiocytosis with advanced lung involvement. J Radiol Case Rep 6 (2012): 22-28.
- Tazi A, Soler P, Hance AJ. Adult pulmonary Langerhans’ cell histiocytosis. Thorax 55 (2000): 405-416.
- Abbott GF, Rosado-de-Christenson ML, Frazier AA, et al. Lymphangioleiomyomatosis: radiologic-pathologic correlation 1. Radiographics 25 (2005): 803-828.
- Abdallah M, Gen´ ereau T, Donadieu J, et al. Langerhans’ ´ cell histiocytosis of the liver in adults. Clinics and Research in Hepatology and Gastroenterology 35 (2011): 475-481.


 Impact Factor: * 5.31
Impact Factor: * 5.31 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
