Appendicitis: How It has Changed Over the Last Few Decades
Leonard Swischuk1*, Dai Chung2, Hal K Hawkins3, Suimin Qiu4, Ravi Radhakrishnan5, Sifrance Tran6, Alvin Camacho7
1Professor of Radiology and Pediatrics, Director, Pediatric Radiology, University of Texas Medical Branch, Galveston, Texas, USA
2Professor in the Department of Surgery, Chief of the Division of Pediatric Surgery, UT Southwestern Medical Center, Dallas, Texas, USA
3Professor, Department of Pediatrics Member, Graduate School of Biomedical Sciences Electron Microscopy Laboratory, University of Texas Medical Brach, Galveston, Texas, USA
4Professor, Department of Pathology, Division of Surgical Pathology, University of Texas Medical Brach, Galveston, Texas, USA
5Associate Professor and Chief, Pediatric Surgery, University of Texas Medical Branch, Galveston, Texas, USA 6Assistant Professor, Pediatric Surgery, University of Texas Medical Branch, Galveston, Texas, USA
7Assistant Professor of Radiology, University of Texas Medical Branch, Galveston, Texas, USA
*Corresponding Author: Dr. Leonard Swischuk, Professor of Radiology and Pediatrics, Director, Pediatric Radiology, Radiology Residency/Fellowship Director, University of Texas Medical Branch, Galveston, Texas, USA
Received: 23 August 2019; Accepted: 16 September 2019; Published: 26 November 2019
Article Information
Citation: Leonard Swischuk, Dai Chung, Hal K Hawkins, Suimin Qiu, Ravi Radhakrishnan, Sifrance Tran, Alvin Camacho. Appendicitis: How It has Changed Over the Last Few Decades. Archives of Clinical and Medical Case Reports 3 (2019): 621-631.
View / Download Pdf Share at FacebookKeywords
<p>Appendicitis; Lymphoid Hyperplasia</p>
Article Details
1. Introduction
For as long as medicine has been around, appendicitis has been considered to be caused by a fecalith obstructing the appendiceal lumen. However, over the years it has come to be noticed by us that a fecalith was not always present and eventually were able to document that it was present in approximately 50% of patients [1]. In the other patients, appendicitis was secondary to appendiceal lymphoid hyperplasia leading to ischemic necrosis of the appendix, especially the appendiceal tip, and subsequent purulent appendicitis [1]. Our first encounter with this type of appendicitis was in a case where the imaging findings were not clearly those of purulent appendicitis but the patient had enough concerning clinical findings to be taken to surgery and Dr. Dai Chung, the surgeon at that time, obtained a photograph of the appendix and came to me and said “Look - this is what we call a pink appendix” (Figure 1). The appendix was grossly swollen, but the serosa was intact. However, it was markedly injected. There was no purulent exudate and no purple (ischemic/vascular) insult to the appendix. In addition, the head of the appendix was slightly enlarged and prominent. We did not notice this finding initially but now have come to realize that it is commonly seen with the pink appendix, and is dealt with later. At that time, we did not know what we were looking at but as it turned out we were looking at an enlarged, inflamed appendix secondary to viral induced lymphoid hyperplasia. This should not have been surprising for the normal appendix has lymphoid tissue in its makeup and for the most part behaves as do lymph nodes throughout the body. In other words it responds to infection, including viral infection and the reactive hyperplastic lymphoid tissue results in a swollen appendix, the “pink appendix” of viral appendicitis.
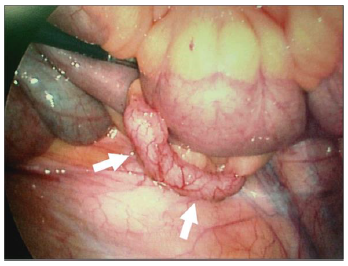
Figure 1: Pink appendix. Note the swollen rounded appendix (arrows) with serosal vascular congestion all leading to the pink appendix. Reprinted with permission from reference article number 14.
The concept of lymphoid hyperplasia being associated with appendicitis is not new and there have been many publications addressing the subject directly or indirectly [1-12]. Although, none of these communications came to a definite conclusion about the relationship of lymphoid hyperplasia to appendicitis the predominate theory was that lymphoid hyperplasia could cause obstruction of the appendix and subsequent purulent appendicitis. More recently an article by Sheridan et al. [13] focused more clearly on this problem and very much coincided with our publication on appendicitis without a fecalith [14], and so, with all of this it is becoming more clear that lymphoid hyperplasia is involved in a good many cases of appendicitis. However, it probably is not so much that it produces obstruction of the appendiceal lumen as that it compromises blood flow to the appendix, especially the tip of the appendix. However, before going into a more detailed discussion of all this it might be worthwhile to take a look at typical purulent appendicitis.
With both ultrasound and CT, the appendix in purulent appendicitis is swollen up to 8-9 millimeters or more. It is fluid filled, has a thickended wall, shows periappendiceal edema, and has a fecalith (Figure 2). When compared with the normal appendix there is no question as to the difference of the two entities. The normal appendix usually measures 4 millimeters or less in diameter, is collapsed to a variable degree, and has no periappendiceal fluid/edema or significant intraluminal fluid (Figure 3). On gross pathology the appendix of classic purulent appendicitis is swollen, with scattered areas of exudate over the surface and some areas of destroyed serosa. It is usually dark red or purple in color (Figure 4), very different from the normal appendix. Furthermore the normal appendix and the one with purulent appendicitis have different findings than those seen with the “Pink Appendix”.
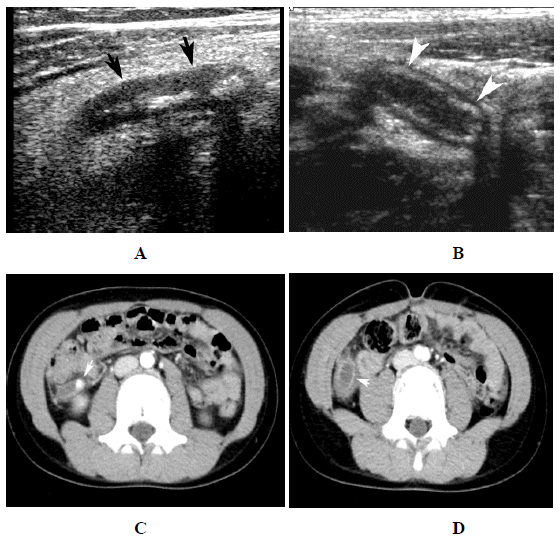
Figure 2: Purulent appendicitis: ultrasound and CT imaging. A. Note the swollen hypodense appendix (arrows). Echogenic edema surrounds the appendix and two fecalith are seen within the appendix. B. Another patient with a fluid filled enlarged appendix (arrow heads). C. CT study demonstrating a fecalith (arrows) in an inflamed appendix. D. A slightly deeper image demonstrates the swollen thick walled appendix full of fluid. Edema is present around the appendix.
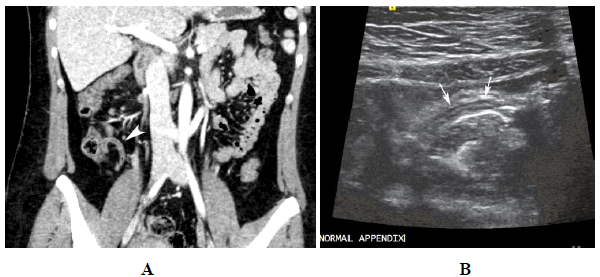
Figure 3: Normal appendix. A. CT study demonstrates a normal, thin appendix (arrow head). B. Ultrasound study demonstrates a hypodense thin appendix (arrows) with an echogenic stripe within it. This is the appendiceal lumen.
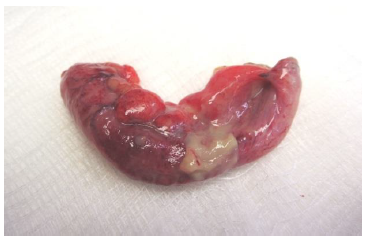
Figure 4: Purulent appendicitis: gross specimen. Note the swollen appendix with exudate over the surface of its midpoint. It is dark red and somewhat purple. All of this reflects hemorrhage and purulent exudate in a grossly inflamed appendix.
In terms of the gross and micro pathology of the pink appendix, it is swollen and circumferentially round. It does not have any serosal bleeding, necrosis, or exudate and in very mild forms the wall is nearly white while with more pronounced involvement it becomes variably pink from serosal vascular injection (Figure 5A and B). It is the serosal vascular injection which leads to the appendix being pink and for the most part these appendices do not need to be removed. On gross histologic examination, they demonstrate lymphoid tissue in the form of a variably thick peripheral intra-appendiceal purple ring (Figure 5C). These appendices usually measures up to 6mm in diameter and it is at this point that the appendix becomes full of lymphoid tissue (Figure 6A) and squeezes the appendiceal lumen to a fine line on ultrasonic graphic longitudinal imaging and a small central dot on cross-sectional imaging (Figure 6B and C). The ultrasound spectrum of findings from the normal appendix to an early pink appendix and then a full blown pink appendix is demonstrated in (Figure 6D).
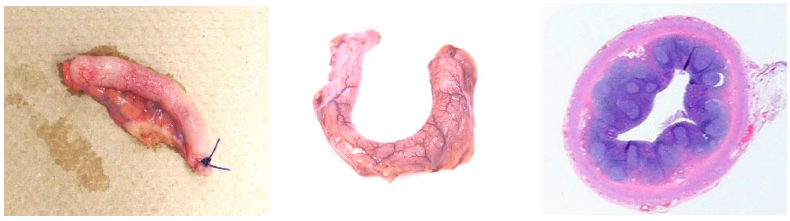
Figure 5: Pink appendix: gross specimens. A. Note the relatively thin and mild pink appendix. It is almost white but slightly pink. B. A more advanced case demonstrates more pronounced serosal vascular congestion and mild prominence of the appendiceal tip but no signs of hemorrhage, necrosis, or exudate. C. Low powered cross section of pink appendix. Note the inner purple layer which consists of lymphoid tissue. The round intact germinal centers are readily visualized.
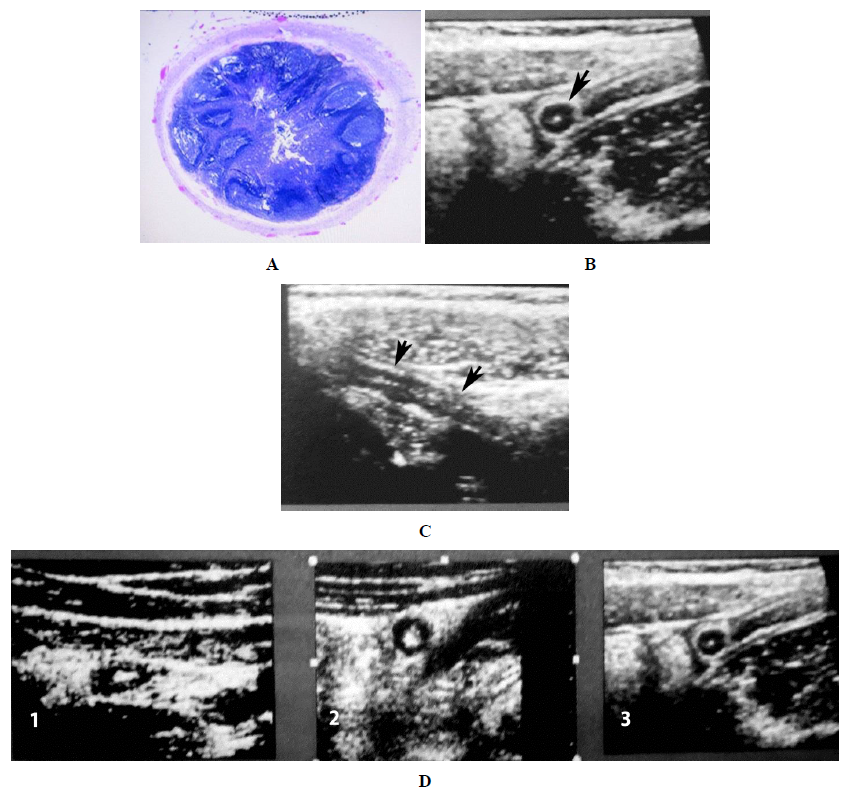
Figure 6: Pink appendix: Uncomplicated. A. Cross-section of a pink appendix shows vascular congestion of the serosal surface and numerous lymphocytic germinal centers filling the lumen of the appendix. There is no hemorrhage. B. Ultrasound cross section. Note the hypodense round appendix (arrows) with a central white dot representing the compressed lumen. There is no edema around the appendix. C. Same patient, longitudinal view demonstrated the same hypodense appendix (arrows) with a central irregular line running through it, again the compressed lumen. D. Sequence of normal to pink appendix: [1] Normal appendix is oval and measures no more than 4mm in diameter. The echogenic lumen is oval. [2] Early lymphoid hyperplasia makes the appendix more round. The lumen is still sizable but becoming round [3]. Full stage pink appendix shows lymphoid tissue filling the appendix with a central echogenic dot representing the compressed lumen. Figure 6B and D reproduced with permission from reference article number 14.
On CT examination, the pink appendix also is round and shows a slightly thickened/hyperintense wall (Figure 7). The center is hypodense and lucent and at first, this hypodense center was thought to be fluid but once we became familiar with the ultrasound findings it was clear that the hypodense center represented lymphoid tissue. This is not a surprise as lymphoid tissue is predominantly fluid and these appendices generally do not need to be removed. However, they can go on to purulent appendicitis and this problem is not rare. Purulent appendicitis can develop in the mid appendix but most often is the tip that is involved. In these patients, in the early stages the tip of the pink appendix is variably swollen and fuzzy because there is now more inflammation present (Figure 8). The diameter is more than 6 mm and all of this occurs because of compromised blood flow to the tip of the appendix which in terms of blood supply is most vulnerable. What happens in these cases is that as the lymphoid tissue fills the appendix the constricting serosa leads to a “compartment syndrome” which then squeezes the blood vessels and causes decreased flow. Since the tip is most vulnerable, it is usually the first part of the appendix to suffer from this problem.
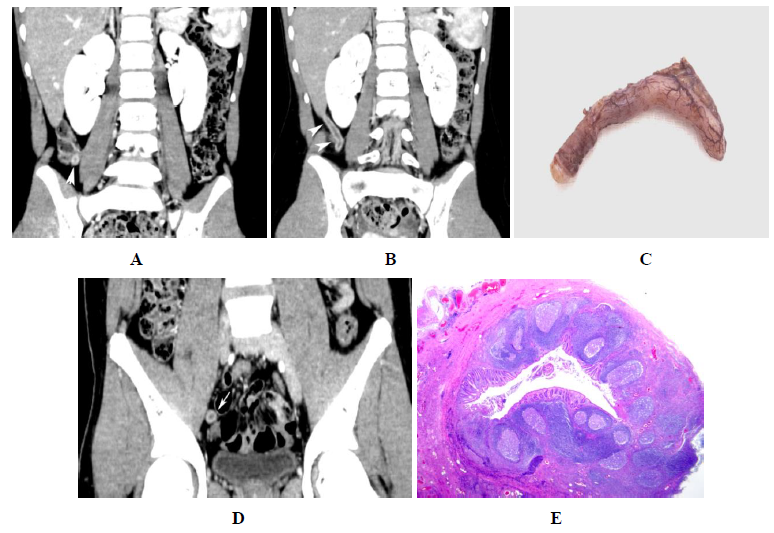
Figure 7: Pink appendix: CT Imaging. A. Note the round appendix (arrows) with a dense wall and central lucency representing lymphoid tissue (arrows). The outer white ring is the wall of the appendix which is exaggerated with CT imaging. There is no edema around the appendix. B. Longitudinal view shows the same appendix (arrows) C. Gross pathologic specimen of the same appendix. D. Another patient with a typical cross-sectional image of a pink appendix (arrow) E. Gross histopathologic image of the same appendix. Note the abundant lymphoid tissue with numerous germinal centers.
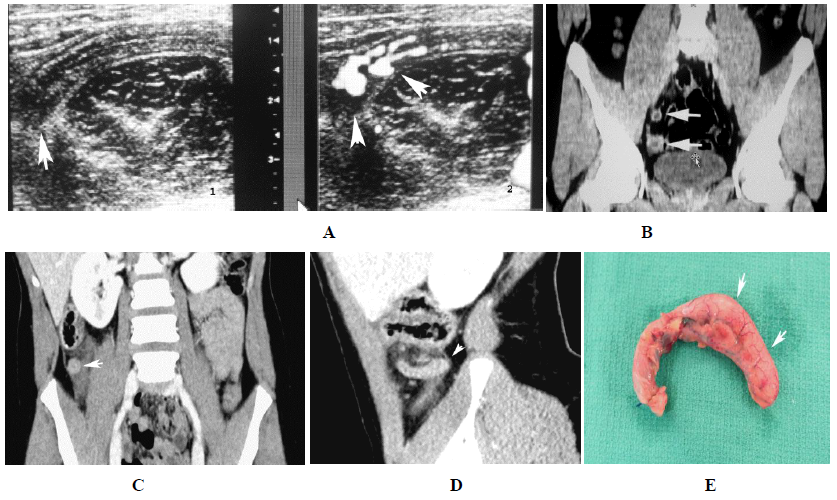
Figure 8: Pink appendix: Purulent tip. A. On image number 1 of this ultrasound study the tip is enlarged and is beginning to be destroyed (arrow). The remainder of the appendix appears as a normal pink appendix. On image 2 with color added there is increased vascularity and flow to the tip of the appendix (arrows). B. CT study in another patient. The upper arrow demonstrates the normal pink appendix. The lower arrow demonstrates the swollen tip. There is no real edema around it but it is swollen. C. In this patient the tip of the appendix is a little swollen and indistinct (arrow). D. Same patient, longitudinal image demonstrating the swollen tip (arrow). E. Gross specimen of the appendix. Note the swollen intact tip (arrows) and the more normal appearing proximal third or so of the pink appendix. Figure A, B, C, and D reproduced with permission from reference article number 14.
Histologically in patients with appendicitis of the tip of the appendix the appendiceal tip shows disorganization of the germinal centers along with hemorrhage and necrosis (Figure 9). For the most part these appendices need to be removed but in some cases, they can regress. Therefore, it is absolutely imperative that one obtain a surgical consultation for in these cases if the surgical consultation says that the patient is a surgical candidate then the patient goes straight to surgery. If it is assessed that the patient is not a surgical candidate then the patient must be observed in-house for 23 hours. By that time the patient will be cleared one way or another. The patient will be able to go home or the patient will go to surgery.
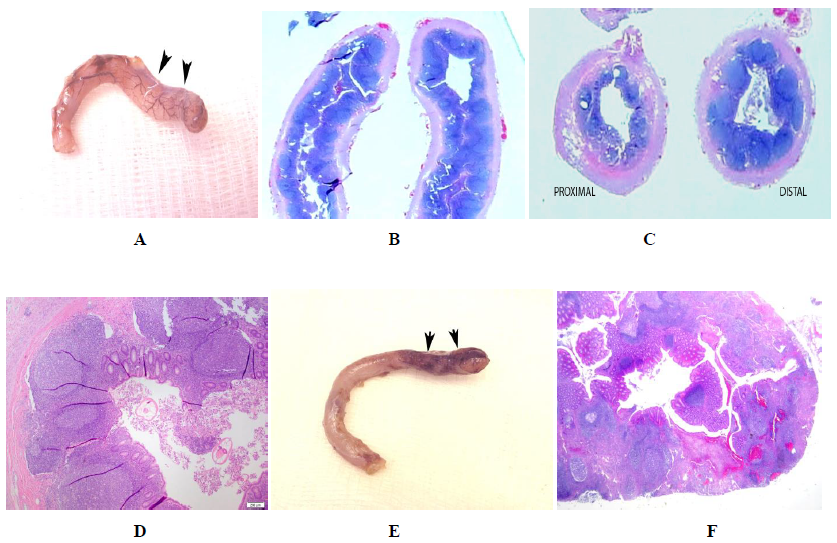
Figure 9: Pink appendix with purulent tip: Histologic features. A. Note that the tip of this pink appendix is swollen (arrows) B. Longitudinal gross pathologic slide of the appendiceal tip demonstrates an abundance of lymphoid tissue with relatively intact germinal centers. C. Same patient. Proximally [proximal] there is very little lymphoid tissue. Distally distal through the swollen part of the appendix there is abundant lymphoid tissue. D. Microscopic evaluation of the same patient demonstrates a number of germinal centers which are still relatively intact but beginning to disorganize. E. Another appendix with a definite purulent tip (arrows). Note that the remainder of the appendix is a normal pink appendix. F. Low power microscopic examination of the tip demonstrates disorganized, destroyed germinal centers and hemorrhage.
In the past when these pink appendices were encountered they were considered normal and therefore the diameter of the normal appendix was pushed from 4 mm to 6 mm. However, once it became clear that the pink appendix was not “classically” normal but that it was a distinct entity which most often measured around 6 mm, the 4 mm diameter of the normal appendix was brought back into play.
Finally, there are those cases where a fecalith is present along with lymphoid hyperplasia. What happens in these cases is that the fecalith likely has been there for months or maybe years and then all of the sudden lymphoid hyperplasia arrives on the scene and forms a collar around the fecalith. There does not have to be a lot of lymphoid tissue to form an obstructing collar. This then totally occludes the appendiceal lumen and purulent appendicitis results (Figure 10).
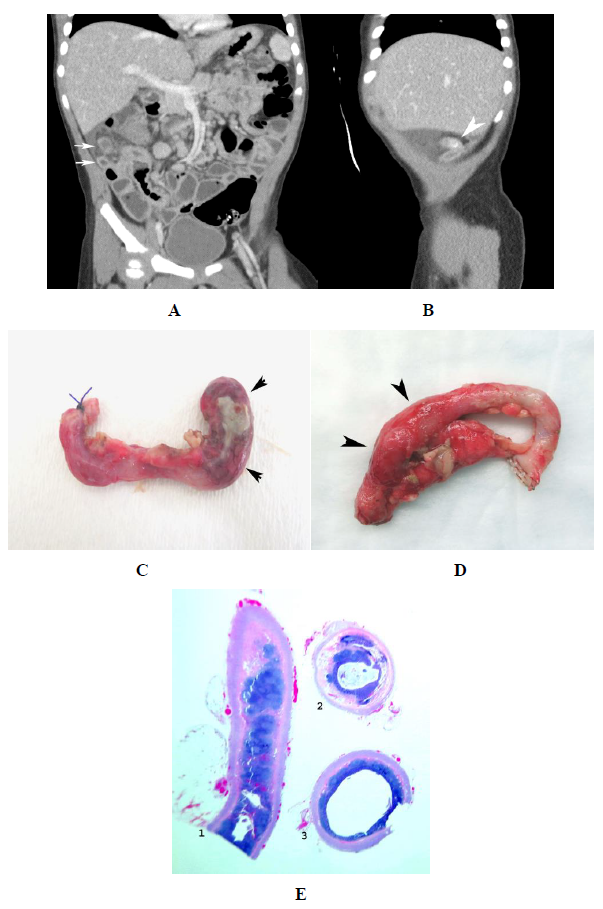
Figure 10: Pink appendix with fecalith. A. Note that the tip of the appendix (upper arrow) is swollen, indistinct and has surrounding edema. The lower arrow points to the more proximal appendix which looks more like a normal pink appendix. B. Same patient demonstrating a fecalith (arrow) in the middle of the appendix. C. Gross appendiceal study demonstrates the tip of the appendix (arrows) which was distal to the location of the fecalith to show typical purulent appendicitis with exudate. D. Another patient with similar findings on gross histologic examination of the appendix. The distal half is swollen (arrow heads) and the proximal half appears more like a normal pink appendix. E. Gross pathology: [1] longitudinal section through the appendiceal tip demonstrates serosal injection, abundance of lymphoid tissue with destruction and disorganization of the germinal centers and hemorrhage [2]. Proximal appendix. Note that there is very little lymphoid tissue [3]. Center of the appendix where the fecalith was present. Note the thin collar of purple lymhiod tissue. It is this collar which surrounded the fecalith and led to appendiceal lumen obstruction.
In summary, if the pink appendix measures 6mm or less and has a clear sharp edge it most likely will settle down and not need removal. However, once this appendix measures more than 6 mm and the edge of the appendix becomes fuzzy one needs to consider possible surgery and obtain a surgical consultation. The reason for this is while some if these patients may have their symptoms regress and recover spontaneously, others will go on to purulent appendicitis of the tip.
Disclaimers
None of the authors have anything to claim.
References
- Bohrod MG. The pathogenesis of acute appendicitis. AmJ of Clin Path 16 (1946): 752-760.
- Kissane JM, SmiJh MG. Pathology of infuncy and childhood. The C.V. Mosby Co. Sl Louis, MO (1975): 231.
- Tobe T. Inapparrent virus infection as a trigger of appendicitis. Lancet 1 (1965): l34J-1346.
- Viral appmdicitis. Hum Pathol 12 (1981): 19J-196.
- Grynspan D, Rabah R. Adenoviral appendicitis presenting clinically as acute appendicitis. Pediatr and Develop Pathol 11 (2008): 138-141.
- Lopez-Navidad A, Domingo P, Cadafalcb J, et al. Acute appendicitis complicating infectious mononucleosis: case report and review. Reviews of Infec Dis 12 (1990): 297.
- Lin JL, Bleiweiss IJ, Mendelson MH, et al. Cystomegalovirus-associated appendicitis in a patient with the acquired immunodeficiency syndrome. Amer J Medicine 89 (1990): 377.
- Neumayer LA, Makar R, Ampel NM, et al. Cytomegalovirus appendicitis in a patient with human immunodeficieocy virus infection. Arch Surg I 28 (1993): 167.
- Lamps LW. Infectious causes of appendicitis. Infect Dis Clin North Am 24 (2010): 995-1018.
- Kelly CS, Kelly RE Jr. Lymphadenopathy in children. Pediatr Clin North Am 45 (1998): 875-888.
- Couture A, Baud C. Gastrointestinal Tract Sonography in Fetuses and Children. Berlin, Germany: Springer (2008).
- Park NH, Oh HE, Park HJ, et al. Ultrasonography of normal and abnormal appendix in children. Worlk J Radiol 3 (2011): 85-91.
- Alison D. Sheridan, Lauren Ehrlich, Rajfaella A. Marotti, et al. Sonographic Distinction Between Acute Suppurative Appendicitis and Viral Appendiceal Lymphoid Hyperplasia ("Pink Appendix") with Pathological Correlation Ultrasound Quarterly Ultrasound Quarterly 31 (2015): 95-98.
- Leonard E. Swischuk, Dai H. Chung, Hal K. Hawkins, et al. Non-fecalith-induced appendicitis: etiology, imaging, and pathology Emerg Radiolo 22 (2015): 643-649.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
