Cryptogenic Organising Pneumonitis: A Clinical Case of A Recurrent Pneumonia
Cátia Pereira*, Joana Coelho, Joana Cardoso, Joana Paixão, Sandra Domingues Santos, Joana Cascais Costa
Centro Hospitalar Universitário de Coimbra, Rua Irmã Lúcia nº2, 3º esquerdo frente; 3030-491, Coimbra, Portugal
*Corresponding Author: Dr. Cátia Pereira, Centro Hospitalar Universitário de Coimbra, Rua Irmã Lúcia nº2, 3º esquerdo frente, 3030-491, Coimbra, Portugal
Received: 23 July 2019; Accepted: 14 August 2019; Published: 15 November 2019
Article Information
Citation: Pereira C, Coelho J, Cardoso J, Paixão J, Santos SD, Costa JC. Cryptogenic Organising Pneumonitis: A Clinical Case of A Recurrent Pneumonia. Archives of Clinical and Medical Case Reports 3 (2019): 457-462.
View / Download Pdf Share at FacebookAbstract
Organising pneumonia (OP) is a clinical, radiological and histopathological entity with unknown etiology, previously named bronchiolitis obliterans with organising pneumonia. A 74-year-old female, presented with asthenia and dyspnea. In the three days before she was on levofloxacin without benefit. She had a previous history of breast cancer and arterial fibrillation under daily amiodarone. During the year before she was three times hospitalized with pneumonia with a weak response to treatments and relapse after apparent resolution. She was re-hospitalized with community acquired pneumonia (CAP) diagnosis having, again, poor response to antibiotics. As there were no clinical or radiological improvements, a chest computed tomography (CT) scan was performed showing diffused pulmonary condensations with air bronchogram. Cryptogenic organizing pneumonia (COP) diagnosis was confirmed through a transbronchial lung biopsy (TLB). This diagnosis difficulty was increased by the interference of concomitant amiodarone intake or even by paraneoplastic hypothesis due to previous breast cancer history, which had to be promptly excluded. Corticotherapy allowed sequential radiological pattern normalization, clinical and analytical improvement. In recurrent pneumonia cases, COP must be considered and further diagnostic tools should be used.
Keywords
<p>Pneumonia; Bronchiolitis Obliterans; Cryptogenic Organising Pneumonia; Corticosteroids</p>
Article Details
Abbreviations:
OP - Organising pneumonia; CAP - Community acquired pneumonia; CT - Chest computed tomography; COP - Cryptogenic organizing pneumonia; TLB - Transbronchial lung biopsy; WBC - White Blood Cell Count1. Introduction
The OP is due to the alveolar epithelium destruction with monocytes and macrophages peribronchial infiltration and airways obstruction [1, 2, 5-9]. Several etiologic factors have been suggested such as viral infections, medication, gastroesophageal reflux, radiotherapy and connectivopathies [3, 7]. It is called COP or idiopathic if all these etiologies have been ruled out [1, 2]. Low fever, cough and dyspnea are the more common symptoms, usually appearing in the 6th decade [6, 10]; considering this nonspecific symptoms, the mean time from the first presentation to diagnosis is about 90 days [6]. Radiologically diffuse, bilateral and asymmetric consolidations are seen in 70-77% of cases and a chest CT scan may reveal asymmetrical consolidations with ground glass appearance [7, 11], extremely useful particularly in cases in which biopsy cannot be performed or is negative [11]. Bronchoalveolar lavage usually demonstrates an elevation of CD8 lymphocytes [6, 12] and TLB may fail to reveal diagnosis. This diagnostic tools have together 86% of sensitivity and should always be performed [11]. The surgical biopsy with thoracoscopy may be necessary [5, 11]. Differential diagnosis with other clinical entities such as CAP is challenging as OP diagnosis does not always present confirmatory histology and though clinical judgment is the mainstay for diagnosis [5, 7, 10]. Symptoms rapidly disappear with corticosteroids introduction, but may return after treatment discontinuation [9, 12]. Corticosteroid therapy can provide recovery in up to 80% of patients in a few weeks to 3 months; although there is no consensus, but doses between 0.75 or 1.5 mg/kg/day have been suggested and should be reduced over weeks to months [7, 8, 10]. Studies have reported the successful use of macrolides in some refractory or recurrent cases [12, 13]. Rituximab, azathioprine and cyclophosphamide may also be used in select cases [7]. In case of a secondary cause of OP was detected, it should be treated according to the underlying cause [6]. Nevertheless relapse is frequent, may reach 58% and is related to late treatment and alkaline phosphatase and gamma-glutamyltransferase elevation [10, 14]. Regarding to recurrence, radiological changes may be more important than clinical findings [14].
2. Case Report
The authors present a case of an 82-year-old female with a history of asthenia for minimal efforts and dyspnea. In the three days before she was on levofloxacin for a respiratory infection. Her past medical history includes chronic stage 3 renal failure, anemia, ischemic cardiac insufficiency, paroxistic auricular fibrillation, obesity, hypertension and history of breast cancer submitted to mastectomy followed by hormonotherapy. She was medicated with furosemide, rivaroxaban, bisoprolol, valsartan and amiodarone. In the past year she was hospitalized three times with pneumonia, each time with slow response to antibiotics. In the last hospitalization, she was treated with levofloxacin; then, and presuming a nosocomial infection, begun piperacilin-tazobactan and vancomicyn switched to meropenem due to poor clinical and analytical improvement. At this point, a contrast enhanced chest CT scan was performed and revealed an air bronchogram and ground glass appearance (Figure 1). Subsequently, the patient was discharged with trimethoprim-sulfamethoxazole. Radiologically, there were only small differences between the first and the last performed chest x rays (Figure 2).
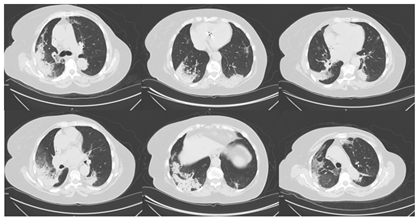
Figure 1: High-resolution computed thorax tomography.
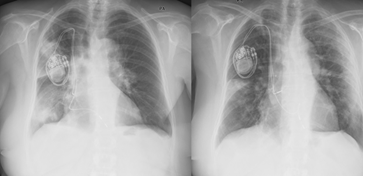
Figure 2: Chest x ray Thorax radiograph before admission and after discharge.
She was hemodynamically stable, with a respiratory rate of 18 cpm, oxygen saturations 94% on room air and temperature of 37.2º Celsius; vesicular murmur was abolished at the right lung base. Her blood results revealed a White Blood Cell Count (WBC) of 11.91 × 103/ml; with a differential of Neutrophils 9.87 × 103/ml and C-reactive protein of 164 mg/l. Serum electrolytes, renal and liver function tests were normal. Her arterial blood gas revealed a pH of 7.6, pO2 68.2 mmHg, pCO2 35.3 mmHg and HCO3 24.8 mmol/l. Sputum cultures, legionella and pneumococcus urinary antigen test were negatives. The chest x ray showed a consolidation both at the right inferior and medium lobe and the left superior lobe (Figure 3). An extended auto-immune lab study was negative. Tumor markers and bacterial and viruses serological tests were also negative. At admission, she was treated with intravenous levofloxacin 750 mg daily, but remained hypoxemic without normalization of infectious parameters. On day 7, levofloxacin was suspended; a chest CT scan was performed, showing diffused pulmonary condensations with air bronchogram and ground glass appearance (Figure 4).
In the attempt to establish a diagnosis, a bronchoscopy was pursued and a bronchial aspirate was obtained, allowing the exclusion of infection or neoplastic cells. Cervical, abdomen and pelvic CT scan demonstrated a fine bilateral pleural effusion. With a presumptive diagnosis of COP, amiodarone was suspended and corticotherapy was initiated at 1 mg/Kg/dia (50 mg). The COP confirmatory diagnosis was obtained with a TBL that revealed an inflammatory infiltrate. The patient showed quick improvement with a sequential radiological evaluation showing radiological pattern normalization (Figure 5).
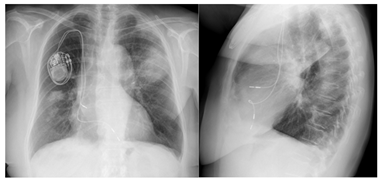
Figure 3: Chest x ray showed consolidation at the right inferior and medium lobe and the left superior lobe.
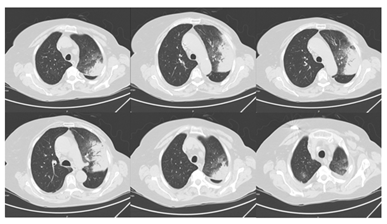
Figure 4: High-resolution computed thorax tomography with diffused pulmonary condensations with air bronchogram and ground glass appearance.
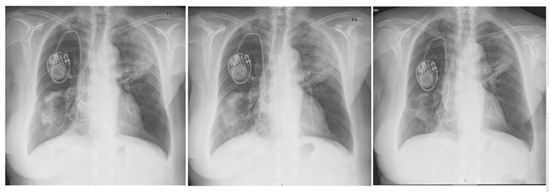
Figure 5: Sequential radiological evaluation showing a radiological pattern normalization.
At the time of hospital discharge, she was prescribed with long term corticosteroid therapy and the mandatory osteoporosis prevention therapy. One month after, she began a well tolerated slowly reduction of corticosteroids dosage. She maintains regular evaluation with Internal Medicine and Pneumology appointments and presently has no respiratory or other relevant complains.
3. Discussion and Conclusions
The OP has several etiologic factors previously referred. In our case report the exclusion of a secondary cause was mandatory because of patient´s amiodarone treatment and the possibility of a paraneoplastic cause. In this case the nonspecific symptoms did not help a rapid diagnosis, radiologically and in the CT scan patient presented diffuse, bilateral and asymmetric consolidations with air bronchogram and ground glass appearance that are frequent in these cases but not totally specifics. The bronchoscopy allowed the exclusion of an infection or neoplastic cause, but the confirmatory diagnosis was obtained with a TBL. A surgical biopsy with thoracoscopy was not performed. Differential diagnosis is extensive. A secondary cause of OP was excluded in this case and with presumptive diagnosis of COP, amiodarone was suspended and corticotherapy was initiated with her clinical improvement. She kept the clinical improvement with corticosteroid therapy, progressive reduction and so we did not consider other alternative therapies. The relapse is frequent but until the moment the patient did not present any disease relapse and maintains her regular medical appointments. This case demonstrates that the absence of clinical improvement under proper antibiotic therapy in a recurrent pneumonia will require broader diagnostic tools in order to clarify the diagnosis.
References
- Baque-Juston M, Pellegrin A, Leroy S, et al. Organizing pneumonia: What is it? A conceptual approach and pictorial review. Diagn Interv Radiol 95 (2014): 771-777.
- Cordier JF. Cryptogenic organising pneumonia. Eur Respir J 28 (2006): 422-446.
- Radzikowska E, Wiatr E, Langfort R, et al. Cryptogenic organizing pneumonia-Results of treatment with clarithromycin versus corticosteroids-Observational study. PLoS ONE 12 (2017): e0184739.
- Pinto-Lopes P, Silva AS, Pimenta J. Non-resolving pneumonia: A case of cryptogenic organizing pneumonia. Porto Biomed. J 1 (2016): 191-192.
- Kang C, Vali Y, Naeem M, et al. Cryptogenic organising pneumonia presenting with spontaneous pneumothorax and the value of procalcitonin: A case report, Respir Med Case Rep 22 (2017): 36-38.
- Vieira AL, Vale A, Melo N, et al. Organizing pneumonia revisited: insights and uncertainties from a series of 67 patients. Sarcoidosis Vasc Diffuse Lung Dis 35 (2018): 129-138.
- Amer K, Walker MC, Yeung V. A Case of Cryptogenic Organizing Pneumonia Managed without a Diagnostic Biopsy. Pulm Med 19 (2018).
- Lee JW, Lee KS, Lee HY, et al. Cryptogenic Organizing Pneumonia: Serial High-Resolution CT Findings in 22 Patients. AJR 195 (2010): 916-922.
- Pearson DJ, Sjulin TJ, Zingalis AA, et al. A Predictive Diagnosis: Organizing Pneumonia. American Journal of Medicine 130 (2017): 917-921.
- Ailing L, Ning X, Tao Q, et al. Successful treatment of suspected organizing pneumonia in a patient without typical imaging and pathological characteristic: A case report. Respir Med Case Rep 22 (2017): 246-250.
- Jara-Palomares L, Gomez-Izquierdo L, Gonzalez-Vergara D, et al. Utility of high-resolution computed tomography and BAL in cryptogenic organizing pneumonia. Respir Med 104 (2010): 1706-1711.
- Ding QL, Lv D, Wang BJ, et al. Macrolide therapy in cryptogenic organizing pneumonia: A case report and literature review. Exp Ther Med 9 (2015): 829-834.
- Ciftci F, Kumbasar O?O?. Seven patients with cryptogenic organizing pneumonia succesfully treated with clarithromycin. Sarcoidosis Vasc Diffuse Lung Dis 35 (2018): 165-170.
- Saito Z, Kaneko Y, Hasegawa T, et al. Predictive factors for relapse of cryptogenic organizing pneumonia. BMC Pulmonary Medicine 19 (2019): 1-6.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
