Endovascular Mechanical Atherothrombectomy (MATH): Using the Rotarex Catheter for Initial Therapy of Acute Lower Limb Ischemia
Miroslav Bulvas MD,PhD*
Department of Interventional Angiology, University Hospital Královské Vinohrady, Surgical Clinic, 3rd School of Medicine, Charles University, Šrobárova, Czech Republic
*Corresponding Author: Dr. Miroslav Bulvas, Department of Interventional Angiology, University Hospital Kralovske Vinohrady, Surgical Clinic, 3rd School of Medicine, Charles University, Srobarova 50, 100 34 Prague, Czech Republic
Received: 05 Aug 2019; Accepted: 30 September 2019; Published: 28 November 2019
Article Information
Citation: Miroslav Bulvas. Endovascular Mechanical Atherothrombectomy (MATH): Using the Rotarex Catheter for Initial Therapy of Acute Lower Limb Ischemia. Archives of Clinical and Medical Case Reports 3 (2019): 660-669.
View / Download Pdf Share at FacebookAbstract
Mechanical atherothrombectomy (MATH) with the Rotarex catheter is rapid, effective and safe technique used as an initial modality for treatment of patients with acute and subacute lower limb ischemia. Contrary to surgery or thrombolysis, this technique has not clinical contraindications and doesn´t require intensive care unit stay. Adoption of MATH has been slow, mainly due to limited experience with the technique and the low standalone efficacy of some devices, neccessitating concomitant use of thrombolysis. The purpose of this review is to present a series of acute arterial occlusions of different anatomy and locations in patients with immediately or marginally threatened extremities.This may help the reader to learn about the technique potential in a vascular emergency states associated with a high mortality and morbidity rates.
Keywords
<p>Clinical; Acute ischemia; Endovascular; Thrombolysis; Cardiopulmonary</p>
Article Details
1. Introduction
Acute ischemia of the lower limbs (ALI) is a critical vascular emergency that not only endangers the affected extremity but also puts the patient‘s life at risk. Catheter-directed thrombolysis (CDT) and/or open surgery (OS) are common therapeutic modalities but both are associated with significant mortality and morbidity rates. Serious comorbidities and cardiopulmonary complications influence postoperational outcome in patients treated by surgery with cardiopulmonary complications responsible for 54%-63% of the deaths at 30 days [1, 2]. On the other hand, the less invasive, thrombolytic treatment is more often complicated by major haemorrhage and stroke [3-5]. As a result, there is no overall difference in limb salvage or death at 30 days, six months or one year after initial surgery and/or initial thrombolysis [6]. A meta-analytic study [6] evaluating data from five randomized trials and 1283 patients reported 30-day mortality of 4.6% (0-12.3%) and 30-day limb salvage of 88% (36-91%) in thrombolysis-treated participants. In surgery patients, the 30-day mortality was 8.2% (4.9-17.5%) and 30-day limb salvage 87% (56-98%). Thrombolytic management was associated with major haemorrhage in 8.8% (0-11.8) of patients at 30 days and in the same group the stroke occured in 1.3% (0-1.8). The stroke did not accompany surgical treatment and major haemorrhage occured in 3.3% (0-5.1). The guidelines recommend surgical therapy [7-10] when the extremity is immediately threatened and time to reperfusion is critical (category II B, early III) [11]. In patients with milder symptoms (category I and II A) [11], thrombolysis is indicated. Due to risk of stroke and major bleeding, certain guidelines [12] recommend surgery over thrombolysis.
The assumption that endovascular therapy without thrombolytics can reduce the number of serious bleeding complications has led to the development of mechanical techniques with the potential for prompt removal of occlusive masses. However, mechanical removal of arterial occlusive material has not replaced the traditional treatments so far. The reasons have been mainly due to limited experience with the techniques and low efficacy of those approaches alone, which often required a combination with thrombolysis and its risk. The Figures 1-6 introduce a different anatomic types that formed a basis of acute lower limb ischemia. Some of them are complex with only a low chance for successful treatment with surgery and /or thrombolysis. The main role of the Rotarex was to remove rapidly the fragmentable occlusive masses (thrombi, emboli, atheromas) and to re-establish the blood flow. The clot removal enabled further treatment of the residual stenoses by the balloon angioplasty (PTA) and/or stenting –stentgrafting (Figures 3, 5, 7). This way, the Rotarex also created the way towards the tibal arteries where the percutaneous aspiration thromboembolectomy, PTA and stenting could be used to enhance the runoff (Figures 1, 3).
2. The Rotarex Device General Description
The Rotarex (Straub Medical AG, Wangs, Switzerland) is a single lumen catheter incorporating a stainless steel helix rotating at up to 60,000 rpm. The Rotarex catheters are equipped with a rotating head driven by the helix, and comprises two superimposed stainless steel cylinders, each with two lateral openings; the outer cylinder is connected to the rotating helix, and the inner cylinder to the catheter shaft. The shape of the head facilitates the detachment and fragmentation of the occlusive material together with a strong vortex created by rotation. The displaced occlusion fragments are aspirated through head openings, and, during that passage, are shredded again into debris that is transported to an external collecting bag. Adjunctive techniques (balloon angioplasty, stenting, percutaneous aspiration thromboembolectomy) can be used to treat underlying (residual) lesions after debulking. Lumen diameter >3 mm and intraluminal passage of the guidewire are the conditions necessary for the Rotarex usage.
3. Cases Characteristics
3.1 Case 1
A female patient with immediately threatened right lower limb by acute ischemia lasting six hours. The primary cause of ischemia was embolism. She was admitted to the hospital with rest pain, sensory loss detectable above the toe level and moderate motor deficit. Groin arterial pulses were palpable with puls deficit distally, Doppler signals from ankle arteries were not audible and the value of Ankle-Brachial Index (ABI) was 0. Mottled skin was cooler compared to contralateral extremity. Intravenous anticoagulation (heparin, 100 unit per kg of weight) was started before hospital admittance. Treatment effect: acute ischemic symptoms disappeared, ABI was 0.81, compartment syndrome appeared temporarily without necessity for fasciotomy. The Rotarex 8F run-time: 4 min., number of catheter passages: 2, intervention total time: 120 min. Additional reintervention or amputation were not necessary during 12 months follow-up. The patient was chronically medicated with Warfarin after the treatment. Medical history: hypercholesterolemia, coronary heart disease, paroxysmal atrial fibrillation (Figure 1).
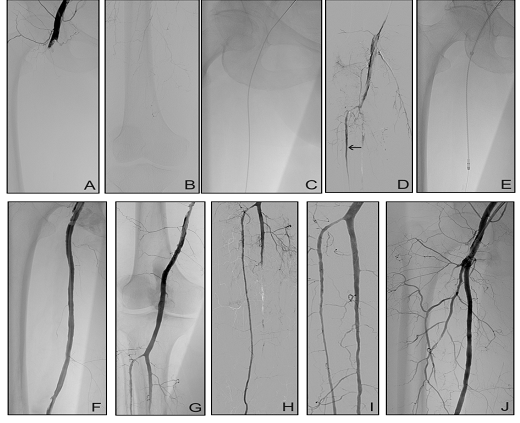
Figure 1: 73-year -old woman, ALI IIb, (A) Common femoral artery occlusion and no visible peripheral vessels on angiography. (B) Introduction of the angiographic catheter. (C) Selective angiography of thrombosed popliteal artery. (D) Patent segment of the tibial anterior artery (arrow). (E) Passage of the Rotarex. (F) Debulked superficial femoral artery (SFA). (G) Patent popliteal artery. (H) Tibial anterior artery is patent to its periphery. (I) Peroneal artery and (J) deep femoral artery (DFA) were recanalized by adjunctive percutaneous aspiration thromboembolectomy (PAT).
3.2 Case 2
A female patient with immediately threatened left lower extremity due to acute ischemia caused by embolism with symptoms duration of 3 hours. The symptoms included rest pain, sensorimotor deficit with pale and cold skin, ABI-0. Intravenous heparin was administered before admittance to the hospital. Left groin arterial pulses were not palpable and therefore contralateral, retrograde femoral puncture was used for angiography and catheter treatment. Four passes of the Rotarex catheter were necessary with total Rotarex 8F run-time: 1 minute, intervention total time: 45 min. Treatment effect: symptoms of acute ischemia disappeared, ABI increased to 0.88. Reintervention or amputation were not necessary during 12 months follow-up. The patient was chronically medicated with Warfarin after the treatment. Medical history: diabetes mellitus, breast cancer, ileofemoral venous thrombosis, pulmonary hypertension, atrial septum defect, brain attack (Figure 2).
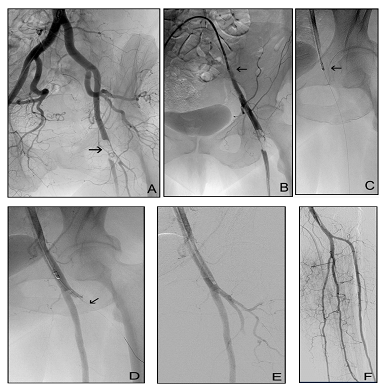
Figure 2: 70- year-old woman, ALI IIb. (A) Left common femoral artery embolism (arrow). (B) Percutaneous sheath (arrow) was introduced from contralateral approach. (C) First, debulking of SFA was performed with the Rotarex (arrow). (D) Subsequently, DFA occlusion (arrow) was traversed with guidewire. (E) Final result. (F) Runoff angiography.
3.3 Case 3
A male patient with acute left lower limb ischemia and symptoms worsening during 24 hours. The problem was initiated by squatting. At the moment of hospital admittance, the patient presented with extremity rest pain, pale and cold skin, sensory loss and moderate motor deficit. Left groin arterial pulses were palpable but a puls deficit was detected more distally. Doppler signals from ankle arteries were not audible and the value of ABI was 0. Intravenous heparin infusion was initiated before the admittance. Intervention total time was 60 minutes with 2 pasages of the Rotarex 8F catheter and its total run-time 1 minute. After intervention, acute ischemic symptoms disappeared, ABI increased to 1.0, and fasciotomy was necessary because of compartment syndrome. After 12-month follow-up, the patient lived with good function of both lower limbs and slowly healing skin defect after the fasciotomy. A patent contralateral popliteal aneurysm was managed surgically and oral anticoagulation with warfarin was chronically administered. Medical history: smoking, hypercholesterolemia (Figure 3).
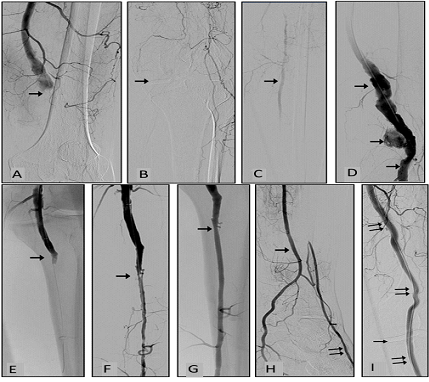
Figure 3: 56-year-old man, ALI IIb, popliteal aneurysm thrombosis and calf vessels occlusions. (A) Digital subtraction angiography (DSA), the arrow indicates beginning of popliteal artery occlusion. (B) Knee joint aperture is depicted by arrow. (C) Just one fragment (arrow) of the calf vessel is filled with contrast material via collaterals. (D) Situation after mechanical debulking of popliteal artery (arrows). (E) Popliteal artery was recanalized (arrow) with the Rotarex catheter. (F) Popliteal artery debulking opened the way for successful (PAT) of peroneal artery with residual stenosis (arrow). (G) Stenosis after PTA and stenting (arrow). (H) Peroneal artery (arrow) supplies plantar vessels directly and dorsal pedis artery (double arrow) via collaterals. (I) Finally, popliteal aneurysm was eliminated by covered stents (double arrows). Single arrow indicates the knee joint aperture.
3.4 Case 4
A female patient admitted to the hospital for sudden onset of bilateral lower limb ischemia lasting 1 hour. The sensorimotor deficit was profound bilaterally with cool and pale skin. Contact with the patient was worsened because of chronic cerebrovascular disease and brain degenerative process associated with long-term walking inability. Arterial pulsations were not detectable on palpation and audible by Doppler (ABI-0), the Duplex examination found both common femoral arteries patent. The foot surface veins collapsed. Intravenous heparin (100 units per kg of weight) was administered before hospital admission. Retrograde approaches from common femoral arteries were used for angiography and endovascular treatment with the Rotarex 8F. Five passes of the catheter and 2.5 minutes of the total run-time were necessary for recanalization without adjunctive percutaneous modalities. Intervention total time: 45 min. The patient was placed on Intensive Care Unit where she presented with prolonged episode of mild systemic hypotension requiring intravenous nutrition and hydration. Chronic renal incompetence temporarily worsened after the contrast material injected during endovascular procedure. Post-intervention period was complicated by bilateral compartment syndrome which did not require fasciotomy. She was discharged from hospital 19 days later to the home for elderly with prolonged health care, without symptoms of lower limb ischemia. Chronic administration of low molecular weight heparin was recommended. Later, she was lost from follow-up. Medical history: hypertension, hypercholesterolemia, recurrent brain attacks, coronary heart disease with episodes of supraventricular arrhythmias including atrial fibrillation, chronic renal incompetence, diabetes (Figure 4).
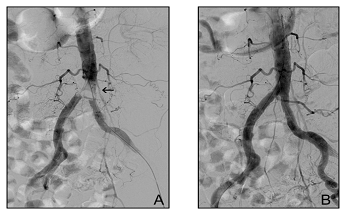
Figure 4: 81-year-old woman, ALI IIb-III. (A) Aortic bifurcation saddle embolism (arrow). (B) Angiography after embolus fragmentation and its aspiration with the Rotarex from the left groin. Balloon catheter stayed inflated in the right common iliac artery during the Rotarex run to protect the right extremity from potential embolism.
3.5 Case 5
A male patient with subacute ischemia of the left lower limb and gradual shortening of claudication interval to 100 m during 2 months caused by in-stent thrombosis. Clinically, he was classified as category 3 (severe claudication) of the Rutherford classification for chronic ischemia [11] before treatment. Calf and foot skin of the affected extremity presented reduced hair and temperature compared to contralateral limb. Calf muscles volume and consistency were reduced and softer on palpation. Groin arterial pulses were palpable with no audible murmur or palpable swirl and ABI was 0.3. Endovascular intervention started with antegrade puncture of the left common femoral artery. Three passages and 1.5 min run-time of the Rotarex 8F were necessary for arterial recanalization followed by Drug Coated Balloon angioplasty and short stenting of residual stenosis. Intervention total time was 45 min. ABI increased to 1.0 and the Rutherford category improved to 0 (no claudication) after the intervention. During 12-month follow-up, clopidogrel (75 mg) and Aspirin (100 mg) were administered daily as a chronic antiaggregation therapy without reappearance of claudication or reintervention. The in-stent reocclusion of the left superficial femoral artery occured after next 17 months and it was successfully managed by the Rotarex again. Medical history: smoking, hypercholesterolemia, hypertension, carotid artery disease, peripheral vascular occlusive disease of the left lower limb with the superficial femoral artery stenting 3 years ago (and subsequent chronic Aspirin administration, 75 mg/d) (Figure 5).
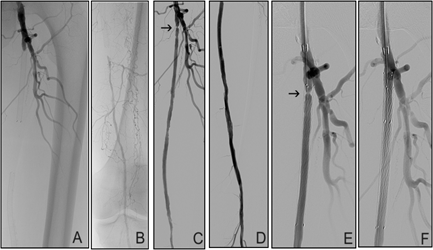
Figure 5: 67-year-old man, ALI IIa. (A)+(B) In-stent occlusion of SFA. (C)+(D) Recanalized lumen after the Rotarex debulking. (C) + (E)) Residual stenosis (arrow). (F) Stenosis correction (adjunctive PTA + stenting).
3.6 Case 6
A man with acute right lower limb ischemia and the thigh claudication (100 m) lasting 48 hours as the first clinical sign of peripheral vascular disease. He presented with mild toe sensor deficit, absent rest pain, adequate skin temperature and well filled foot skin veins. Femoral and peripheral arterial pulses were not palpable and the murmur was audible above the right iliac artery in rest. ABI was 0.62 before treatment. Contralateral approach was used for angiography and endovascular treatment with the Rotarex 8F catheter. Three catheter passages were needed for recanalization, the total run-time was 1.5 minutes and intervention total time: 65 min. Acute ischemia symptoms disappeared after the therapy and the Ankle-Brachial Index increased to 1.0. Clopidogrel was administered as a chronic antiaggregation therapy and the patient is asymptomatic for 6 years. Medical history: smoking (Figure 6).
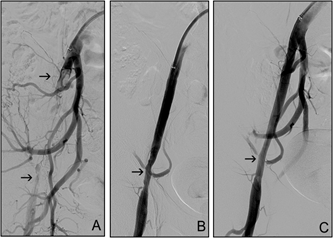
Figure 6: 45-year-old man, ALI IIa, external iliac artery occlusion. (A) Acute thrombosis is located between arrows. (B) Residual stenosis after debulking. (C) Angiogram after adjunctive PTA.
4. Literature Data
The technical success varied from 92% to 100% in previous studies with target occlusions in infraaortic arteries [13], femoropopliteal arterial segments [14-18] and femoropopliteal bypasses [19, 20]. Lower technical success was reported in subgroups with occluded bypass grafts (78%) and crossover approach (56%) [13]. Frequency of concomitant thrombolytic therapy varied between 0% [18] and 14% [19]. Number of surgical revascularization procedures ranged from 0% [13, 15, 17-21] to 5.3% [14] and mortality rate varied between 0 [14, 15, 18] and 1% [13, 17] at 30 days. Thirty days clinical success varied from 68% to 98%, secondary patency from 68% to 97.6%, amputation-free survival from 94.4% to 100%, frequency of major complications from 0% to 6.9%, major haemorrhage from 0% to 2.6% and frequency of major debulking device-related complications from 0% to 0.4%. After the therapy, significant elevation of mean ABI was regularly reported.
5. Conclusions
Mechanical debulking with the Rotarex offers an eligible initial therapeutic technique for patients with acute and subacute ischemia of lower limbs. It allows safe removal of occlusive material, prompt revascularization without major distant haemorrhage and without the need to place the patient in an intensive care unit. With low invasiveness, prompt reperfusion, chance for immediate treatment of underlying and concomitant lesions, together with a low rate of bleeding complications, mechanical debulking offers advantages over catheter-directed thrombolysis and vascular surgery. Debulking with the Rotarex also opens an access to infrapopliteal vessels and enables to enhance the runoff during the same procedure.
References
- de Marino PM, López IM, Suero SR, et al. Results of infrainguinal bypass in acute limb ischaemia. Eur J Vasc Endovasc Surg 51 (2016): 824-830.
- Rush DS, Frame SB, Bell RM, et al. Does open fasciotomy contribute to morbidity and mortality after acute lower extremity ischemia and revascularization? J Vasc Surg 10 (1989): 343-356.
- Ouriel K, Veith FJ, Sasahara AA. A comparison of recombinant urokinase with vascular surgery as initial treatment for acute arterial occlusion of the legs. Thrombolysis or Peripheral Arterial Surgery (TOPAS) Investigators. N Engl J Med 338 (1998): 1105-1111.
- Palfreyman SJ, Booth A, Michaels JA. A systematic review of intra-arterial thrombolytic therapy for lower-limb ischaemia. Eur J Vasc Endovasc Surg 19 (2000): 143-157.
- Earnshaw JJ, Whitman B, Foy C. National Audit of Thrombolysis for Acute Leg Ischemia (NATALI): clinical factors associated with early outcome. J Vasc Surg 39 (2004): 1018-1025.
- Berridge DC, Kessel DO, Robertson I. Surgery versus thrombolysis for initial management of acute limb ischaemia. Cochrane Database of Systematic Reviews 6 (2013): 1-32.
- Rajan DK, Patel NH, Valji K, et al. Quality improvement guidelines for percutaneous management of acute limb ischemia. J Vasc Interv Radiol 20 (2009): S208-S218.
- Norgren L, Hiatt WR, Dormandy JA, et al. Inter-society consensus for the management of peripheral arterial disease (TASC II). Eur J Vasc Endovasc Surg 33 (2007): S1-S75.
- Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): executive summary. A collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease). Endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation. J Am Coll Cardiol 47 (2006): 1239-1312.
- Tendera M, Aboyans V, Bartelink ML, et al. ESC Guidelines on the diagnosis and treatment of peripheral artery diseases: document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries. The task force on the diagnosis and treatment of peripheral artery diseases of the European Society of Cardiology (ESC). European Heart Journal 32 (2011): 2851-2906.
- Rutherford RB, Baker JD, Ernst C, et al. Recommended standards for reports dealing with lower extremity ischemia: revised version. J Vasc Surg 26 (1997): 517-538.
- Alonso-Coello P, Bellmunt S, McGorrian C, et al. Antithrombotic therapy in peripheral artery disease: antithrombotic therapy and prevention of thrombosis, 9th Edn: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141 (2012): e669S.
- Zeller T, Frank U, Bürgelin K, et al. Early experience with a rotational thrombectomy device for treatment of acute and subacute infra-aortic arterial occlusions. J Endovasc Ther 10 (2003): 322-331.
- Duc SR, Schoch E, Pfyffer M, et al. Recanalization of acute and subacute femoropoplital artery occlusions with the Rotarex catheter: one year follow-up, single center experience. Cardiovasc Intervent Radiol 28 (2005): 603-610.
- Wissgott C, Kamusella P, Richter A, et al. Mechanical rotational thrombectomy for treatment of acute and subacute occlusions of femoropopliteal arteries: retrospective analysis of the results from 1999 to 2005. Röfo 180 (2008): 325-331.
- Stan?k F, Ouhrabková R, Procházka D. Mechanical thrombectomy using the Rotarex catheter in the treatment of acute and subacute occlusions of peripheral arteries: immediate results, long-term follow-up. Int Angiol 32 (2013): 52-60.
- Freitas B, Steiner S, Bausback Y, et al. Rotarex mechanical debulking in acute and subacute arterial lesions: single-center experience with 525 patients. Angiology 68 (2017): 233-241.
- Bérczi V, Deutschmann HA, Schedlbauer P, et al. Early experience and midterm follow-up results with a new, rotational thrombectomy catheter. Cardiovasc Intervent Radiol 25 (2002): 275-281.
- Lichtenberg M, Käunicke M, Hailer B. Percutaneous mechanical thrombectomy for treatment of acute femoropopliteal bypass occlusion. Vascular Health and Risk Management 8 (2012): 283-289.
- Wissgott C, Kamusella P, Andresen R. Recanalization of acute and subacute venous and synthetic bypass-graft occlusions with mechanical rotational catheter. Cardiovasc Intervent Radiol 36 (2013): 936-942.
- Bulvas M, Sommerová Z, Van?k I, et al. Prospective single-arm trial of debulking as initial therapy in patients with acute and subacute lower limb ischemia: one-year outcomes. J Endovasc Ther 26 (2019): 291-301.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
