Lanosterol Eye Drops in a Human Juvenile Nuclear Cataract
Felici A1*, Mengato D2, Falciani M3 and Bertelli E4
1Department of Geriatrics, Bolzano's Central Public Hospital, Italy
2University of Padova, Department of Pharmaceutical and Pharmacological Sciences Padova, School of Hospital Pharmacy, Italy
3Department of Clinical Pharmacy, Bolzano's Central Public Hospital, Italy
4Department of Ophthalmology, Bolzano's Central Public Hospital, Italy
*Corresponding Author: Dr. Alessandro Felici, Department of Geriatrics, Bolzano's Central Public Hospital, Italy, Tel: +39 0471 908111;
Received: 26 December 2017; Accepted: 15 January 2018; Published: 18 January 2018
Article Information
View / Download Pdf Share at FacebookAbstract
A revolutionary non-surgical and low-cost treatment of the largest cause of world's blindness could have an immeasurable effect on global health, since surgical procedures are not accessible to all patients worldwide. In cataracts we still keep replacing a clouded lens without considering the pathogenetic mechanism of lens clouding, and without helping the lens epithelium to restore its homeostatic functions. In the light of recent scientific findings and considering the lack of studies on humans in this field we tested lanosterol eye drops on a human juvenile nuclear cataract with a lower lanosterol concentration and with a different vehicle compared to the eye drops recently tested on animals. Our experiment has demonstrated that there were no clinically relevant changes in intraocular pressure neither reversal of cataract using eye drops with a relatively low dose of lanosterol. These results encourage to test eye drops with higher lanosterol concentrations on human cataract cases.
Keywords
Blindness, Lens clouding, Human cataract, Lanosterol
Article Details
1. Introduction
Cataract is considered responsible for the half the world's blindness and 33% of visual impairment worldwide [1, 2] and a nonsurgical treatment would potentially be able to prevent cataract formation and to treat cataracts without the surgical costs and risks also in developing countries, where cataracts contribute to more than 90% of the total disability-adjusted life year [2] as well as in countries where access to eye care is limited. Actually the protein aggregation amidst the regularly arranged crystallins of lens fibers is considered responsible of the pathogenesis of cataract [3], and the finding of mutations in lanosterol synthase as a cause for cataract formation in rats and subsequently in families with congenital cataract has encouraged in evaluating its role in the formation of senile cataract in rabbits and dogs, where Zhao et al. have shown that lanosterol acts by reversing the protein aggregation within the lens fibers both in vitro and in vivo [4]. In order to study the effects of lanosterol in human cataract we evaluated if a lanosterol 5 mM solution could reverse a unilateral juvenile nuclear cataract.
2. Subject and Methods
An evaluation request was asked to the Hospital Ethical Committee before preparing the eye drops and informed consent was obtained from the patient. Lanosterol 5mM solution was prepared by adding Ph. Eur. sterile olive oil to lanosterol 20 mg powder (Sigma Aldrich) at room temperature to a final volume of 8 ml. Because of the solubility grade of lanosterol in olive oil, the mixture needed more than 20 minutes of manual dissolution. After that we performed a filtration (with a 0.2 µm filter designed for oily solutions) to guarantee appropriated sterility to the eye drops. The patient with idiopathic unilateral juvenile nuclear cataract was treated with lanosterol 5 mM eye drops two times daily for the first week and three times daily for the next seven weeks. Visual acuity, intraocular pressure and slit lamp examination were repeatedly measured during this period.
3. Results
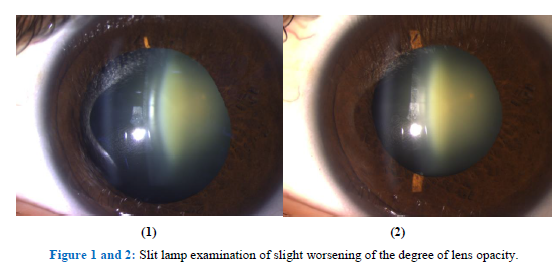
During the study period the patient showed a progressive worsening of visual acuity with an increasing of miopic shift (Table 1) as well as a slight worsening of the degree of lens opacity in slit lamp examination (See Figure 1 and 2), without clinically relevant effects on intra-ocular pressure during the whole observation period (Table 2).
4. Conclusion
Lanosterol 5 mM oily ophtalmic solution did not improve visual acuity in a single case of human idiopathic juvenile nuclear cataract.
Time |
Myopic Shift |
|
Visit 1 |
-0.5 |
|
Visit 2 |
-3 |
|
Visit 3 |
-3.5 |
|
Visit 4 |
-4 |
|
Visit 5 |
-13 |
Table 1: The patient showed a progressive worsening of visual acuity with an increasing of miopic shift.
|
|
Intraocular Pressure |
|
|
Time |
Affected Eye |
Contralateral Eye |
|
Visit 1 |
19 |
16 |
|
Visit 2 |
19 |
17 |
|
Visit 3 |
18 |
11 |
|
Visit 4 |
19 |
14 |
|
Visit 5 |
17 |
14 |
Table 2: Effects on intra-ocular pressure during the whole observation period.
5. Discussion
Before testing lanosterol eye drops on humans we reconsidered the eye drops composition of the lanosterol eye drops recently tested on animals in order to have a potentially more appropriate vehicle for human use. Sterile olive oil was the vehicle chosen because of the moderate solubility of lanosterol in it that permitted to avoid the use of ethanol (EtOH) in order to have an expected greater tolerability. A relatively low concentration of lanosterol was chosen because of the lack of studies on humans in this field. In our experiment five mM lanosterol solution failed to reverse nuclear opacity of human juvenile nuclear cataract after 8 weeks of treatment. During the study period no cataract regression, no visual acuity improvement and no visual acuity stabilization could be observed. However the concentration of lanosterol used by us may have been inadequate to reverse the nuclear cataract in human lenses. Shanmugam et al. found that Twenty-five mM lanosterol solution failed to reverse nuclear opacity of human age-related cataractous nuclei after 6 days of incubation [5]. Considering that it is also possible that molecules other than lanosterol could play a role in human nuclear cataract reversal, specially in age-related senile cataracts, where a variety of biochemical and physical insults lead to changes in the lens proteins, determining increased protein aggregation and cataract formation. Hejtmancik et al. have elucidated that congenital and senile cataracts show different molecular pathways [6, 7], in particular mutations in enzymes such as lanosterol synthase and or every mutation that destabilize the lens proteins lead to congenital cataract formation due to increased protein aggregation. Furthermore, multiple authors have shown that congenital cataracts are usually secondary to missense mutations causing the accumulation of altered protein residues or, in specific cases, to ferritin levels, while age-related imbalance of chaperones and accumulation of degraded and denatured normal proteins was usually shown in senile cataract [8]. Therefore, future basic science research and clinical studies may provide us with more information concerning different pathogenesis and more specific treatments. It may be reasonable to verify if different lanosterol concentrations or the choice of a different solvent or treating cataract with different etiologies could improve clinical outcomes.
References
- Rao GN, Khanna R, Payal A. The global burden of cataract. Curr Opin Ophthalmol 22 (2011): 4-9.
- Pascolini D, Mariotti SPM. Global estimates of visual impairment: 2010. British Journal Ophthalmology (2011).
- Moreau KL, King JA. Protein misfolding and aggregation in cataract disease and prospects for prevention. Trends Mol Med 18 (2012): 273-282.
- Zhao L, Chen XJ, Zhu J, et al. Lanosterol reverses protein aggregation in cataracts. Nature 523 (2015): 607-11.
- Shanmugam PM, Barigali A, Kadaskar J, et al. Effect of lanosterol on human cataract nucleus. Indian J Ophthalmol 63 (2015): 888-890.
- Hejtmancik JF. Congenital cataracts and their molecular genetics. Semin Cell Dev Biol 19 (2008): 134-149.
- Hejtmancik JF, Kantorow M. Molecular genetics of age-related cataract. Exp Eye Res 79 (2004): 3-9.
- Beaumont C, Leneuve P, Devaux I, et al. Mutation in the iron responsive element of the L ferritin mRNA in a family with dominant hyperferritinaemia and cataract. Nat Genet 11 (1995): 444-446.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
