Two Cases of Pneumocystis In Patients Treated with Osimertinib
Foch Emilie*, Masse Laurie, Allou Nathalie, Moreau Diane, Andre Michel
Pneumologie, Centre Hospitalier Universitaire Felix Guyon, Allée des Topazes, Saint Denis, France
*Corresponding Author: Foch Emilie, Pneumologie, Centre Hospitalier Universitaire Felix Guyon, Allée des Topazes, Saint Denis, France
Received: 21 January 2020; Accepted: 14 April 2020; Published: 24 May 2021
Article Information
Citation: Foch Emilie, Masse Laurie, Allou Nathalie, Moreau Diane, Andre Michel. Two Cases of Pneumocystis In Patients Treated with Osimertinib. Archives of Clinical and Medical Case Reports 5 (2021): 479-481.
View / Download Pdf Share at FacebookKeywords
<p>Lung adenocarcinoma; Osimertinib; Pneumocystis</p>
Article Details
1. Background
The first-line treatment in patients with advanced lung adenocarcinoma and EGFR-activating mutation is osimertinib 80 mg/day [1]. In patients with advanced adenocarcinoma of the lung and EGFR exon 20 mutation, osimertinib 160 mg/day can be given off-label after at least one line of treatment [2]. Here we report two cases of pneumocystis in patients treated with osimertinib.
2. Case Report
Patient 1 was a 78-year-old patient treated for stage IVB adenocarcinoma of the lung with EGFR-activating mutation. First-line treatment with osimertinib 80mg/day was initiated on day 10 (12/26/2019), followed by osimertinib 40 mg/day on day 52 (renal and cutaneous side effects were observed). The patient was hospitalized on day 162 for dyspnea. A thoracic CT scan showed ground-glass interstitial lung disease (Figure 1). Biological results confirmed the presence of grade 2 lymphopenia (with a nadir of 0.7 G/L), which had appeared at day 40. Bronchoalveolar lavage was positive by PCR for Pneumocystis carinii. HIV serology was negative. The patient was treated with corticosteroids 1mg/kg with gradual decrease and cotrimoxazole, but developed digestive and cutaneous side effects. She was then treated with wellvone, which led to clinical and radiological improvement (Figure 1).
Patient 2 was a 76-year-old patient treated for stage IVA adenocarcinoma of the lung with EGFR exon 20 mutation. Third-line treatment with osimertinib 160 mg/day was initiated on day 53. The patient was hospitalized on day 84 for dyspnea. A thoracic CT scan showed ground-glass interstitial lung disease (Figure 2). Biological results showed grade 0 lymphopenia (with a nadir of 1.09 G/L), which was attributed to the use of osimertinib. Induced sputum was positive by PCR for Pneumocystis carinii. HIV serology was negative. The patient was treated with corticosteroids 1 mg/kg with gradual decrease and cotrimoxazole, but developed neurological side effects. She was then treated with atovaquone, which led to clinical and radiological improvement (Figure 2).
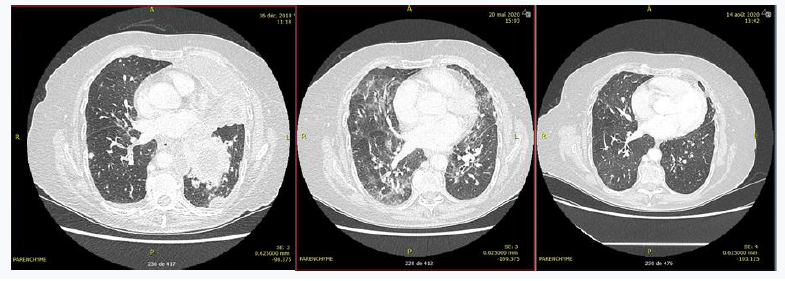
Figure 1: Parenchymal CT scan of Patient 1 before treatment with osimertinib (day 1), before anti-pneumocystis treatment (day 156), and after anti-pneumocystis treatment (day 242)
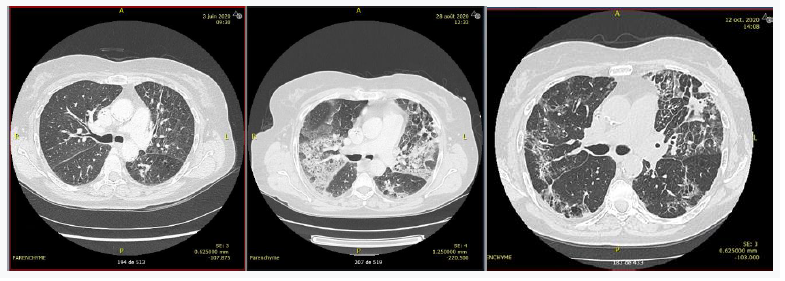
Figure 2: Parenchymal CT scan of Patient 2 before treatment with osimertinib (day 1), before anti-pneumocystis treatment (day 86), and after anti-pneumocystis treatment (day 131).
3. Discussion
In the FLAURA and AURA studies (AURA3, AURAex, AURA2, and AURA1), lymphopenia as a side effect of osimertinib was found in 67% of included patients, of whom 7.2% had grade 3 lymphopenia [3]. No other cases of pneumocystis in patients treated with osimertinib were found in the literature or in public databases [4-5]. These two cases raise the question of prophylaxis for pneumocystis in patients with lymphopenia due to osimertinib.
References
- Soria J-C, Ohe Y, Vansteenkiste J, et al. Osimertinib in Untreated EGFR-Mutated Advanced Non-Small-Cell Lung Cancer. N Engl J Med 378 (2018): 113-125.
- Piotrowska Z, et al. - ASCO® 2020 – Abs.#9513.
- Summary of product characteristics – Osimertinib.
- Public drug database. available at http://basedonnees-publique.medicaments.gouv.fr/
- Hematox®. available at http://www.biourtox.com/Mediquick7/Abstract.cfm?id_dci=1268


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
